9.6 Surface tension
Surface Tension

Surface tension is the tendency of liquid surfaces to shrink to the minimum surface area due to cohesive forces.
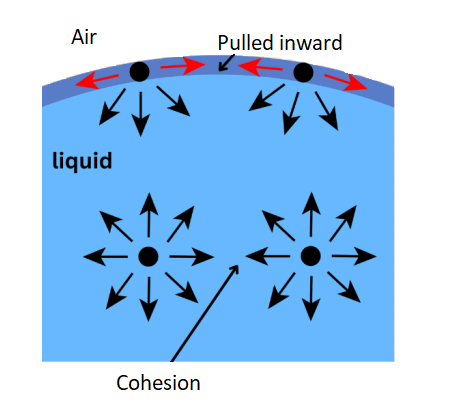
- Bulk molecules are pulled from all directions. A molecule in the bulk liquid experiences cohesive forces with other molecules in all directions.
- Surface molecules are pulled inward. A molecule at the surface of a liquid experiences only net inward cohesive forces.
A microscopic view of water illustrates the difference between molecules at the surface of a liquid and water molecules within a liquid. A small drop of liquid is spherical in shape, because for given volume, sphere has minimum surface area. This stretched behaviour is due to net downward force acting on a molecule on the surface of liquid as shown in the figure below. The cohesive forces between molecules down into a liquid are shared with all neighboring atoms. Those on the surface have no neighboring atoms above and exhibit stronger attractive forces upon their nearest neighbors on the surface. This enhancement of the intermolecular attractive forces at the surface is called surface tension.

For molecules at the surface, the cohesive forces are unbalanced as there are no molecules above them. This means molecules at the surface of a liquid are pulled towards their neighbours below the surface. This creates a “skin” at the surface known as surface tension.
A molecule well inside the liquid experiences no net force (the one that is submerged).
Now, consider a line \(A B\) on the free surface of the liquid. The small elements of the surface on this line are in equilibrium because they are acted upon by equal and opposite forces, acting perpendicular to the line from either side as shown in the figure below.

The force acting on this line is proportional to the length of this line. If \(l\) is the length of imaginary line and \(F\) the total force on either side of the line,
\(F \propto l\)
\(
S=T=\frac{F}{l}
\)
where:
\(S\) is the surface tension,
\(F\) is the force acting perpendicular to the line, and \(l\) is the length of the line.
Surface tension, \(S=\frac{\text { Force }}{\text { Length }}\)
From this expression, surface tension can be defined as the force acting per unit length of an imaginary line drawn on the liquid surface, the direction of force being perpendicular to this line and tangential to the liquid surface.
It is denoted by \(S\) and it is a scalar quantity.
Units and dimensions of surface tension
SI unit of surface tension \(=\mathrm{Nm}^{-1}\), CGS unit of surface tension \(=\) dyne \(\mathrm{cm}^{-1}\),
Dimensions of surface tension
\(
=\frac{\text { Force }}{\text { Length }}=\frac{\left[\mathrm{MLT}^{-2}\right]}{[\mathrm{L}]}=\left[\mathrm{ML}^0 \mathrm{~T}^{-2}\right]
\)
Examples of Surface tension Shown below:
The Molecular Cause (Cohesion): At the molecular level, water molecules are attracted to each other by hydrogen bonds.
Inside the liquid: A molecule is pulled in all directions by its neighbors, so the net force is zero.
At the surface: There are no water molecules above the surface to pull upward. Consequently, the molecules at the top are pulled inward and sideways, creating a “skin” or tension that resists being broken.
Formation of Spherical Raindrops:
Surface tension acts to minimize the surface area of a liquid. For a given volume, a sphere has the smallest possible surface area. This is why falling water or small droplets on a leaf always pull themselves into a round shape.
Water strider, which are small insects, can walk on water as their weight is considerably less to penetrate the water surface. Like this, there are various examples of surface tension which are found in nature. Some cases are provided below:

Example 1: A liquid is kept in a beaker of radius 4 cm . Consider a diameter of the beaker on the surface of the water. Find the force by which the surface on one side of the diameter pulls the surface on the other side. (Take, surface tension of liquid \(=0.075 \mathrm{Nm}^{-1}\) )
Solution: The length of the diameter, \(l=2 r=8 \mathrm{~cm}=0.08 \mathrm{~m}\)
The surface tension is \(S=F / l\).
Thus, \(F=S l=\left(0.075 \mathrm{Nm}^{-1}\right) \times(0.08 \mathrm{~m})=6 \times 10^{-3} \mathrm{~N}\)
Surface energy
The free surface of a liquid always has a tendency to contract and possess minimum surface area. To increase the surface area of the liquid work has to be done. This work done is stored in the surface film of the liquid as its potential energy. This potential energy per unit area of the surface film is called the surface energy.
Hence, the surface energy may be defined as the amount of work done in increasing the area of the surface film through unity. Thus,
\(
\text { Surface energy }=\frac{\text { Work done in increasing the surface area }}{\text { Increase in surface area }}=\frac{W}{\Delta A}
\)
The SI unit of surface energy is \(\mathrm{Jm}^{-2}\).
Relation between surface energy and surface tension

Consider a rectangular frame \(P Q R S\). Here, wire \(Q R\) is movable. A soap film is formed on the frame. The film pulls the movable wire \(Q R\) inward due to surface tension.
As, \(\quad\) surface tension \(=\frac{\text { force }}{\text { length }}=\frac{F^{\prime}}{2 l} \Rightarrow F^{\prime}=S \times 2 l\) (A film has two sides and the liquid in between so length is \(2l\))
Suppose that we move the bar by a small distance \(d\) as shown. Since the area of the surface increases, the system now has more energy, this means that some work has been done against an internal force.
If \(Q R\) is moved through a distance \(d\) by an external force \(F\) very slowly, then some work has to be done against this force.
\(\therefore\) External work done \(=\) Force \(\times\) Distance
\(=S \times 2 l \times d \quad\left(\because F^{\prime}=F\right)\)
Increase in surface area of film \(=2 l \times d\) (As soap film has two sides)
Surface energy \(=\frac{\text { Work done }}{\text { Surface area }}=\frac{S 2 l d}{2 l d}=S\)
So, value of surface energy of liquid is numerically equal to the value of surface tension.
Note:
- Work done in forming a drop, \(W=S \times 4 \pi r^2\)
\(
\left(A s, \Delta A=4 \pi r^2-0=4 \pi r^2\right)
\) - Work done in forming a bubble, \(W=S \times 4 \pi r^2 \times 2\) (As, bubble in air has two surfaces)
Example 2: How much work will be done in increasing the diameter of a soap bubble from 2 cm to 5 cm . (Take, surface tension of soap solution is \(3.0 \times 10^{-2} \mathrm{Nm}^{-2}\) )
Solution: The work done in increasing the diameter of a soap bubble from 2 cm to 5 cm .
What’s given in the problem?
Initial diameter of the soap bubble: \(\boldsymbol{d}_{\mathbf{1}}=2 \mathrm{~cm}=0.02 \mathrm{~m}\)
Final diameter of the soap bubble: \(d_2=5 \mathrm{~cm}=0.05 \mathrm{~m}\)
Surface tension of soap solution: \(S=3.0 \times 10^{-2} \mathrm{Nm}^{-1}\)
The surface area of a sphere is given by \(A=4 \pi r^2\), where \(r\) is the radius.
A soap bubble has two surfaces, so the total surface area is \(2 A=8 \pi r^2\).
The work done in changing the surface area of a soap bubble is given by \(W=S \Delta A\), where \(\Delta A\) is the change in surface area.
Calculate the initial surface area \(A_1\)
\(
r_1=\frac{d_1}{2}
\)
\(
A_1=4 \pi r_1^2
\)
Calculate the final surface area \(\boldsymbol{A}_2\)
\(
r_2=\frac{d_2}{2}
\)
\(
A_2=4 \pi r_2^2
\)
\(
\Delta A=2(A_2-A_1) \text { Soap bubble has two surfaces. }
\)
\(
\begin{aligned}
\Delta A & =2\left[4 \pi\left\{\left(2.5 \times 10^{-2}\right)^2-\left(1.0 \times 10^{-2}\right)^2\right\}\right] \\
& =1.32 \times 10^{-2} \mathrm{~m}^2
\end{aligned}
\)
\(\begin{aligned} \therefore \text { Work done, } W & =\left(3.0 \times 10^{-2}\right)\left(1.32 \times 10^{-2}\right) \mathrm{J} \\ & =3.96 \times 10^{-4} \mathrm{~J}\end{aligned}\)
Example 3: Calculate the energy released when 1000 small water drops each of radius \(10^{-7} \mathrm{~m}\) coalesce to form one large drop. (Take, surface tension of water is \(7.0 \times 10^{-2} \mathrm{Nm}^{-1}\) )
Solution: Let \(r\) be the radius of smaller drops and \(R\) of bigger one. Equating the initial and final volumes, we get
\(
\frac{4}{3} \pi R^3=(1000)\left(\frac{4}{3} \pi r^3\right)
\)
\(
R=10 r=(10)\left(10^{-7}\right) \mathrm{m} \text { or } R=10^{-6} \mathrm{~m}
\)
Further, the water drops have only one free surface.
Therefore,
\(
\begin{aligned}
\Delta A & =4 \pi R^2-(1000)\left(4 \pi r^2\right) \\
& =4 \pi\left[\left(10^{-6}\right)^2-\left(10^3\right)\left(10^{-7}\right)^2\right] \\
& =-36 \pi\left(10^{-12}\right) \mathrm{m}^2
\end{aligned}
\)
Here, negative sign implies that surface area is decreasing. Hence, energy released in the process,
\(
\begin{aligned}
U & =T|\Delta A|=\left(7 \times 10^{-2}\right)\left(36 \pi \times 10^{-12}\right) \mathrm{J} \\
& =7.9 \times 10^{-12} \mathrm{~J}
\end{aligned}
\)
Measuring Surface Tension
A fluid will stick to a solid surface if the surface energy between fluid and the solid is smaller than the sum of surface energies between solid-air, and fluid-air. Now there is attraction between the solid surface and the liquid. It can be directly measured experimentaly as schematically shown in Figure below. A flat vertical glass plate, below which a vessel of some liquid is kept, forms one arm of the balance. The plate is balanced by weights on the other side, with its horizontal edge just over water. The vessel is raised slightly till the liquid just touches the glass plate and pulls it down a little because of surface tension. Weights are added till the plate just clears water.

Suppose the additional weight required is \(W\). Then the surface tension of the liquid-air interface is
\(
S_{l a}=(W / 2 l)=(m g / 2 l)
\)
where \(m\) is the extra mass and \(l\) is the length of the plate edge. The subscript (\(la\)) emphasises the fact that the liquid-air interface tension is involved.
Angle of Contact
When the free surface of a liquid comes in contact with a solid, then the surface of the liquid becomes curved at the point of contact. Whenever the liquid surface becomes a curve, then the angle between the two medium (solid-liquid interface) comes in the picture. Surfaces that repel water, like the leaves of the Colocasia plant (as shown below) , are called hydrophobic. “Hydro-” is a Greek root word that means water, “Phobia” means fear.

When a water drop lands on the Colocasia leaf, it beads up into a sphere and simply rolls away. Why doesn’t the water make the surface wet? Because this leaf is superhydrophobic !

The angle between the tangent to the liquid surface at the point of contact and the solid surface inside the liquid is known as the angle of contact between the solid and the liquid. It is denoted by \(\theta\) (Read it as “theta” which is Greek alphabet small letter).
Its value is different at interfaces of different pairs of solids and liquids. In fact, it is the factor which decides whether a liquid will spread on the surface of a chosen solid or it will form droplets on it.
Let us consider three interfaces such as liquid-air, solid-air and solid-liquid with reference to the point of contact ‘ O ‘ and the interfacial surface tension forces \(\mathrm{T}_{\mathrm{sa}}, \mathrm{T}_{\mathrm{sl}}\) and \(\mathrm{T}_{\mathrm{la}}\) on the respective interfaces as shown in Figure below.

Since the liquid is stable under equilibrium, the surface tension forces between the three interfaces must also be in equilibrium. Therefore,
\(
T_{s a}=T_{l a} \cos \theta+T_{s l} \Rightarrow \cos \theta=\frac{T_{s a}-T_{s l}}{T_{l a}}
\)
From the above equation, there are three different possibilities which can be discussed as follows.
(i) If \(\mathrm{T}_{\mathrm{sa}}>\mathrm{T}_{\mathrm{sl}}\) and \(\mathrm{T}_{\mathrm{sa}}-\mathrm{T}_{\mathrm{sl}}>0\) (water-plastic interface) then the angle of contact \(\theta\) is acute angle ( \(\theta\) less than \(90^{\circ}\) ) as \(\cos \theta\) is positive.
(ii) If \(\mathrm{T}_{\mathrm{sa}}<\mathrm{T}_{\mathrm{sl}}\) and \(\mathrm{T}_{\mathrm{sa}}-\mathrm{T}_{\mathrm{ls}}<0\) (water-leaf interface) then the angle of contact is obtuse angle ( \(\theta\) less than \(180^{\circ}\) ) and as \(\cos \theta\) is negative.
(iii) If \(T_{\mathrm{sa}}>\mathrm{T}_{\mathrm{la}}+\mathrm{T}_{\mathrm{sl}}\) then there will be no equilibrium and liquid will spread over the solid.
Therefore, the concept of angle of contact between the solid-liquid interface leads to some practical applications in real life. For example, soaps and detergents are wetting agents. When they are added to an aqueous solution, they will try to minimize the angle of contact and in turn penetrate well in the cloths and remove the dirt. On the other hand, water proofing paints are coated on the outer side of the building so that it will enhance the angle of contact between the water and the painted surface during the rainfall.
For example, water forms droplets on lotus leaf as shown in Figure below(a) while spreads over a clean plastic plate as shown in Figure below (b).

We consider the three interfacial tensions at all the three interfaces, liquid-air, solid-air and solid-liquid denoted by \(S_{\mathrm{la}}, S_{\mathrm{sa}}\) and \(S_{\mathrm{sl}}\), respectively as given in Fig. (a) and (b). At the line of contact, the surface forces between the three media must be in equilibrium. From the Fig. (b) the following relation is easily derived.
\(
S_{\mathrm{la}} \cos \theta+S_{\mathrm{sl}}=S_{\mathrm{sa}}
\)
The following cases arise:
Case-I: If the surface tension at the solid-liquid \(S_{\mathrm{sl}}\), interface is greater than the surface tension at the liquid-air \(S_{\mathrm{la}}\) interface, i.e. \(S_{\mathrm{sl}}>S_{\mathrm{la}}\), then \(\theta>90^{\circ}\) (the angle of contact is obtuse angle as shown in Fig. (a)).
The molecules of a liquid are attracted strongly to themselves and weakly to those of solid. It costs a lot of energy to create a liquid-solid surface. The liquid then does not wet the solid.
e.g. Water-leaf or glass-mercury interface.
Case-II: If the surface tension at the solid-liquid \(S_{\mathrm{sl}}\) interface is less than the surface tension at the liquid-air \(S_{\text {la }}\) interface, i.e. \(S_{\mathrm{sl}}<S_{\mathrm{la}}\), then \(0<90^{\circ}\) (the angle of contact is acute angle as shown in Fig. (b)).
The molecules of the liquid are strongly attracted to those of solid and weakly attracted to themselves. It costs less energy to create a liquid-solid surface and liquid wets the solid.
e.g. When soap or detergent is added to water, the angle of contact becomes small.
Excess pressure (inside a bubble or drop)
Due to surface tension, a drop or bubble tends to contract and so compresses the matter enclosed. This in turn increases the internal pressure which prevents further contraction and equilibrium is achieved. So, in equilibrium, the pressure inside a bubble or drop is greater than outside and the difference of pressure between two sides of the liquid surface is called excess pressure.
Following ways are used to calculate the excess pressure inside a bubble or drop.
A soap bubble consists of two spherical surface films with a thin layer of liquid between them. Because of surface tension, the film tend to contract in an attempt to minimize their surface area. But as the bubble contracts, it compresses the inside air, eventually increasing the interior pressure to a level that prevents further contraction.

Another interesting consequence of surface tension is that the pressure inside a spherical drop Figure above (a) is more than the pressure outside. Suppose a spherical drop of radius \(r\) is in equilibrium. If its radius increase by \(\Delta r\). The extra surface energy is
\(
\left[4 \pi(r+\Delta r)^2-4 \pi r^2\right] S_{\mathrm{la}}=8 \pi r \Delta r S_{\mathrm{la}}
\)
If the drop is in equilibrium this energy cost is balanced by the energy gain due to expansion under the pressure difference ( \(P_i-P_o\) ) between the inside of the bubble and the outside. The work done is
\(
W=\left(P_i-P_{{o}}\right) 4 \pi r^2 \Delta r
\)
so that
\(
\left(P_{\mathrm{i}}-P_{\mathrm{o}}\right)=\left(2 S_{\mathrm{la}} / r\right)
\)
In general, for a liquid-gas interface, the convex side has a higher pressure than the concave side. For example, an air bubble in a liquid, would have higher pressure inside it. See Figure (b).
A bubble Figure (c) differs from a drop and a cavity; in this it has two interfaces. Applying the above argument we have for a bubble
\(
\Delta P=\left(P_1-P_{\mathrm{o}}\right)=\left(4 S_{\mathrm{la}} / r\right)=\frac{4 T}{r}
\)
This is probably why you have to blow hard, but not too hard, to form a soap bubble. A little extra air pressure is needed inside!
Example 4: 0 .04 cm liquid column balances the excess pressure inside a soap bubble of radius 6 mm . Evaluate density of the liquid. (Take, surface tension of soap solution \(=0.03 \mathrm{Nm}^{-1}\) )
Solution: The excess pressure inside a soap bubble,
\(
\begin{aligned}
\Delta p & =4 T / R \\
& =\frac{4 \times 0.03 \mathrm{Nm}^{-1}}{6 \times 10^{-3} \mathrm{~m}}=20 \mathrm{Nm}^{-2}
\end{aligned}
\)
The pressure due to 0.04 cm of the liquid column,
\(
\Delta p=h \rho g=\left(0.04 \times 10^{-2} \mathrm{~m}\right) \rho\left(10 \mathrm{~ms}^{-2}\right)
\)
Thus, \(20 \mathrm{Nm}^{-2}=\left(0.04 \times 10^{-2} \mathrm{~m}\right) \rho\left(10 \mathrm{~ms}^{-2}\right)\)
\(\therefore\) Density of the liquid, \(\rho=5 \times 10^3 \mathrm{kgm}^{-3}\)
Example 5: Two separate soap bubbles (radii 0.004 m and 0.002 m ) formed of the same liquid (surface tension \(0.07 \mathrm{Nm}^{-1}\) ) come together to form a double bubble. Find the radius and the sense of curvature of the internal film surface common to both the bubbles.
Solution: Excess pressure inside first soap bubble, \(p_1=p_0+\frac{4 T}{r_1}\)
Excess pressure inside second soap bubble, \(p_2=p_0+\frac{4 T}{r_2}\)

\(\because \quad r_2<r_1\), therefore \(p_2>p_1\)
i.e. Pressure inside the smaller bubble will be more. The excess pressure,
\(
p=p_2-p_1=4 T\left(\frac{r_1-r_2}{r_1 r_2}\right) \dots(i)
\)
This excess pressure acts from concave to convex side, the interface will be concave towards smaller bubble and convex towards larger bubble. Let \(R\) be the radius of interface, then
\(
p=\frac{4 T}{R} \dots(ii)
\)
From Eqs. (i) and (ii), we get
\(
R=\frac{r_1 r_2}{r_1-r_2}=\frac{(0.004)(0.002)}{(0.004-0.002)}=0.004 \mathrm{~m}
\)
Note: Excess pressure inside a liquid drop A liquid drop has only one surface film, so excess pressure inside the liquid drop is given by
\(
\Delta p=\frac{2 T}{R}
\)
Some Important points regarding excess pressure
Case-I: If we have an air bubble inside a liquid, a single surface is formed. There is air on the concave side and liquid on the convex side. The pressure in the concave side (i.e. in the air) is greater than the pressure in the convex side (i.e. in the liquid) by an amount \(2 T / R\).

The above expression has been written by assuming \(p_1\) to be constant from all sides of the bubble. For small size bubbles this can be assumed.
Case II: From the above discussion, we can make a general statement. The pressure on the concave side of a spherical liquid surface is greater than the convex side by \(2 T / R\) as shown below.

Case-III: Excess pressure inside a soap bubble can also be understood in terms of excess pressure inside a curved surface as shown below.

\begin{aligned}
&\begin{array}{r}
p_1-p^{\prime}=\frac{2 T}{R} \\
\Rightarrow \quad p^{\prime}-p_2=\frac{2 T}{R}
\end{array}\\
&\text { Excess pressure, } \Delta p=p_1-p_2=\frac{4 T}{R}
\end{aligned}
\)
Case-IV: If two bubbles of different sizes are connected with each other through a thin tube (as shown in figure), then the air will rush from smaller to larger bubble (pressure inside smaller bubble will be greater than pressure inside larger bubble), so that the smaller will shrink while the larger will expand till the smaller bubble reduces to a droplet.

Example 6: What should be the pressure inside a small air bubble of 0.1 mm radius situated just below the water surface? (Take, surface tension of water \(=7.2 \times 10^{-2} \mathrm{Nm}^{-1}\) and atmospheric pressure \(=1.013 \times 10^5 \mathrm{Nm}^{-2}\) )
Solution: Surface tension of water,
\(
T=7.2 \times 10^{-2} \mathrm{Nm}^{-1}
\)
Radius of air bubble,
\(
\begin{aligned}
R & =0.1 \mathrm{~mm} \\
& =10^{-4} \mathrm{~m}
\end{aligned}
\)
The excess pressure inside the air bubble is given by
\(
p_2-p_1=\frac{2 T}{R}
\)
\(\therefore\) Pressure inside the air bubble, \(p_2=p_1+\frac{2 T}{R}\)
Substituting the values, we get
\(
\begin{aligned}
p_2 & =\left(1.013 \times 10^5\right)+\frac{\left(2 \times 7.2 \times 10^{-2}\right)}{10^{-4}} \\
& =1.027 \times 10^5 \mathrm{Nm}^{-2}
\end{aligned}
\)
Shape of liquid surface
The curved surface of the liquid is called meniscus. The shape of the meniscus (convex or concave) is determined by the relative strengths of cohesive and adhesive forces. The force between the molecules of the same material is known as cohesive force and the force between the molecules of different kinds of material is called adhesive force.
When the adhesive force between solid and liquid molecules is more than the cohesive force between liquid-liquid molecules (as with water and glass), shape of the meniscus is concave and the angle of contact \(\theta\) is less than \(90^{\circ}\). In this case, the liquid wets or adheres to the solid surface.

When the adhesive force between solid and liquid molecules is less than the cohesive force between liquid-liquid molecules than shape of meniscus is convex.
Note The angle of contact between water and clean glass is zero and that between mercury and clean glass is \(137^{\circ}\).
Capillarity
The term capilla means hair which is Latin word. A tube of very fine (hair-like) bore is called a capillary tube.

If a capillary tube of glass is dipped in liquid like water, the liquid rises in the tube, but when the capillary tube is dipped in a liquid like mercury, the level of liquid falls in the tube.
This phenomenon of rise or fall of a liquid in the capillary is called capillarity.
Some examples are
(i) Small capillaries in fibres of towels soaks water from our skin.
(ii) In trees sap rises in stem due to capillary action.
Capillary Rise

One consequence of the pressure difference across a curved liquid-air interface is the wellknown effect that water rises up in a narrow tube in spite of gravity. The word capilla means hair in Latin; if the tube were hair thin, the rise would be very large. To see this, consider a vertical capillary tube of circular cross section inserted into an open vessel of water (Figure above). The contact angle between water and glass is acute. Thus the surface of water in the capillary is concave. This means that there is a pressure difference between the two sides of the top surface. This is given by
\(
\begin{aligned}
& \left(P_i-P_o\right)=(2 S / r)=2 S /(a \sec \theta) \\
& =(2 S / a) \cos \theta \dots(i)
\end{aligned}
\)
\(a\) is the radius of the capillary tube itself. and \(r\) is the radius of curvature of the meniscus. The meniscus (the curved top surface of the water) is part of a larger imaginary sphere. \(r\) is the radius of that sphere. Thus the pressure of the water inside the tube, just at the meniscus (air-water interface) is less than the atmospheric pressure. Consider the two points A and B in Figure (a) above. They must be at the same pressure, namely
\(
P_o+h \rho g=P_i=P_A \dots(ii)
\)
where \(\rho\) is the density of water and \(h\) is called the capillary rise [Figure (a)]. Using Eq. (i) and (ii) we have
\(
h \rho g=\left(P_i-P_o\right)=(2 S \cos \theta) / a \dots(iii)
\)
\(
h=\frac{2 S \cos \theta}{r \rho g}
\)
The above equations make it clear that the capillary rise is due to surface tension. It is larger, for a smaller a. Typically it is of the order of a few cm for fine capillaries.
Important Points on Formula of Capillary Rise:
When a capillary tube is dipped in a liquid, then the level of liquid in capillary tube rises or falls w.r.t. free surface of liquid outside the capillary.

This phenomena of rises or fall of liquid is called capillary action.
The formula in capillary motion is
\(
h=\frac{2 T \cos \theta}{r \rho g}
\)
where, \(h=\) height of liquid column rises or falls,
\(r=\) radius of capillary tube,
\(\rho=\) density of liquid,
\(g=\) acceleration due to gravity,
\(\theta=\) angle of contact and \(T=\) surface tension.
The result has following notable features
(i) If the contact angle \(\theta\) is greater than \(90^{\circ}\), the term \(\cos \theta\) becomes negative and hence, \(h\) is negative. The result, then gives the depression of the liquid in the tube.
(ii) Suppose a capillary tube is held vertically in a liquid which has a concave meniscus, then capillary rise is given by
\(
\begin{aligned}
& h=\frac{2 T \cos \theta}{r \rho g}=\frac{2 T}{R \rho g} \quad\left(\because R=\frac{r}{\cos \theta}\right) \\
& h R=\frac{2 T}{\rho g}
\end{aligned}
\)
Note:
(i) If the tube is of a length \(l\) less than \(h(l<h)\), the liquid does not overflow. The angle made by liquid surface with the tube adjusts in such a way that rise will stop when it is equal to length of tube.
(ii) If the tube makes an angle \(\phi\) units with vertical, rise will be
\(
h^{\prime}=\frac{h}{\cos \phi}
\)
Example 7: A capillary tube whose inside radius is 0.5 mm is dipped in water having surface tension \(7.0 \times 10^{-2} \mathrm{Nm}^{-1}\). To what height is the water raised above the normal water level? Angle of contact of water with glass is \(0^{\circ}\). Density of water is \(10^3 \mathrm{~kg} \mathrm{~m}^{-3}\) and \(g=9.8 \mathrm{~ms}^{-2}\).
Solution: Height raised, \(h=\frac{2 T \cos \theta}{r \rho g}\) Substituting the proper values, we get
\(
\begin{aligned}
h & =\frac{(2)\left(7.0 \times 10^{-2}\right) \cos 0^{\circ}}{\left(0.5 \times 10^{-3}\right)\left(10^3\right)(9.8)}=2.86 \times 10^{-2} \mathrm{~m} \\
& =2.86 \mathrm{~cm}
\end{aligned}
\)
Example 8: A glass tube of radius 0.4 mm is dipped vertically in water. Find upto what height, the water will rise in the capillary. If the tube is inclined at an angle of \(60^{\circ}\) with the vertical, how much length of the capillary is occupied by water? (Take, surface tension of water \(=7.0 \times 10^{-2} \mathrm{Nm}^{-1}\), density of water \(=10^3 \mathrm{kgm}^{-3}\) )
Solution: For glass-water, angle of contact, \(\theta=0^{\circ}\).
Now, height of water in capillary,
\(
\begin{aligned}
h & =\frac{2 T \cos \theta}{r \rho g}=\frac{(2)\left(7.0 \times 10^{-2}\right) \cos 0^{\circ}}{\left(0.4 \times 10^{-3}\right)\left(10^3\right)(9.8)} \\
& =3.57 \times 10^{-2} \mathrm{~m} \\
& =3.57 \mathrm{~cm}
\end{aligned}
\)
Length of capillary occupied after tilting it is,
\(
\begin{aligned}
l & =\frac{h}{\cos 60^{\circ}}=\frac{3.57}{1 / 2} \\
& =7.14 \mathrm{~cm}
\end{aligned}
\)
Example 9: The lower end of a capillary tube of diameter 2.00 mm is dipped 8.00 cm below the surface of water in a beaker. What is the pressure required in the tube in order to blow a hemispherical bubble at its end in water? The surface tension of water at temperature of the experiments is \(7.30 \times 10^{-2} \mathrm{Nm}^{-1}\). 1 atmospheric pressure \(=\) \(1.01 \times 10^5 \mathrm{~Pa}\), density of water \(=1000 \mathrm{~kg} / \mathrm{m}^3\), \(\mathrm{g}=9.80 \mathrm{~m} \mathrm{~s}^{-2}\). Also calculate the excess pressure.
Solution: The excess pressure in a bubble of gas in a liquid is given by \(2 S / r\), where \(S\) is the surface tension of the liquid-gas interface. You should note there is only one liquid surface in this case. (For a bubble of liquid in a gas, there are two liquid surfaces, so the formula for excess pressure in that case is \(4 S / r\).) The radius of the bubble is \(r\). Now the pressure outside the bubble \(P_o\) equals atmospheric pressure plus the pressure due to 8.00 cm of water column. That is
\(
\begin{aligned}
P_{\mathrm{o}}=\left(1.01 \times 10^5 \mathrm{~Pa}+0.08 \mathrm{~m}\right. & \times 1000 \mathrm{~kg} \mathrm{~m}^{-3} \\
& \left.\times 9.80 \mathrm{~m} \mathrm{~s}^{-2}\right)
\end{aligned}
\)
\(
=1.01784 \times 10^5 \mathrm{~Pa}
\)
Therefore, the pressure inside the bubble is
\(
\begin{aligned}
P_{\mathrm{i}} & =P_{\mathrm{o}}+2 \mathrm{~S} / \mathrm{r} \\
& =1.01784 \times 10^5 \mathrm{~Pa}+\left(2 \times 7.3 \times 10^{-2} \mathrm{~Pa} \mathrm{~m} / 10^{-3} \mathrm{~m}\right) \\
& =(1.01784+0.00146) \times 10^5 \mathrm{~Pa} \\
& =1.02 \times 10^5 \mathrm{~Pa}
\end{aligned}
\)
where the radius of the bubble is taken to be equal to the radius of the capillary tube, since the bubble is hemispherical! (The answer has been rounded off to three significant figures.) The excess pressure in the bubble is 146 Pa.