9.2 Pressure
Pressure
A sharp needle when pressed against our skin pierces it. Our skin, however, remains intact when a blunt object with a wider contact area (say the back of a spoon) is pressed against it with the same force. Such everyday experiences convince us that both the force and its coverage area are important. Smaller the area on which the force acts, greater is the impact. This impact is known as pressure.
Normal force acting per unit area of a surface is called pressure. It is generally represented by \(p\).

If surface area is \(A\) and normal force acting on the surface is \(F\) (shown in figure), then pressure on the surface is
\(
p=\frac{\text { Normal force }(F)}{\text { Area }(A)}
\)
If area is very small, then \(p=\lim _{\Delta A \rightarrow 0} \frac{\Delta F}{\Delta A}=\frac{d F}{d A}\)
It is clear from the above formula that pressure acting on a surface depends upon the normal force acting on the surface and its area. Thus, force of same magnitude exerts different pressure on different area that is for the same force, if area is lesser, then pressure will be greater.
SI unit of pressure is \(\mathrm{Nm}^{-2}\) or pascal (Pa). Pressure is a scalar quantity and its dimensional formula is \(\left[\mathrm{ML}^{-1} \mathrm{~T}^{-2}\right]\).
Pressure in a fluid
When a fluid (either liquid or gas) is at rest, it exerts a force perpendicular to any surface in contact with it, such as a container wall or a body immersed in the fluid. While the fluid as a whole is at rest, the molecules or atoms that make up the fluid are in motion. These molecules or atoms collide continuously with the walls of container. Pressure of a fluid is due to molecules colliding with the container walls. In Figure below, we can see the pressure acting on all sides of the vessel.

The pressure applied by a fluid can be defined as the magnitude of the normal force (applied by fluid) acting per unit surface area.
If the pressure is same at all points of a finite plane surface with area \(A\), then
\(
p=\frac{F_{\perp}}{A}
\)
where, \(F_{\perp}\) is the normal force on the surface.
Unit of fluid pressure French scientist Pascal contributed many researches in the field of fluid pressure. Thus, in his honour, SI unit of pressure is known as pascal which is denoted by \(\mathrm{Pa} .1 \mathrm{~Pa}=1 \mathrm{Nm}^{-2}\)
One unit used practically in meterology is the bar which is equal to \(10^5 \mathrm{~Pa} .1\) bar \(=10^5 \mathrm{~Pa}\)
When an air filled balloon is immersed inside the water in a vessel, it immediately comes up and floats on water. This shows that water (or liquid) exerts pressure in the upward direction. It is shown in Figure below.

Similarly, liquid pressure acts in lateral sides also. When a bottle having water is pierced on the sides we can see water coming out with a speed as in Figure below. This is because liquid exerts lateral pressure on the walls the container.

Example 1: The two thigh bones (femurs), each of cross-sectional area \(10 \mathrm{~cm}^2\) support the upper part of a human body of mass 40 kg. Estimate the average pressure sustained by the femurs.
Solution: Total cross-sectional area of the femurs is \(A=2 \times 10 \mathrm{~cm}^2=20 \times 10^{-4} \mathrm{~m}^2\). The force acting on them is \(F=40 \mathrm{~kg}\) wt \(=400 \mathrm{~N}\) (taking \(g=10 \mathrm{~m} \mathrm{~s}^{-2}\) ). This force is acting vertically down and hence, normally on the femurs. Thus, the average pressure is
\(
P_{a v}=\frac{F}{A}=2 \times 10^5 \mathrm{Nm}^{-2}
\)
Atmospheric pressure \(\left(p_{a}\right)\)
It is the pressure of the earth’s atmosphere. This changes with weather and elevation. Normal atmospheric pressure at sea level (an average value) is \(1.013 \times 10^5 \mathrm{~Pa}\). Thus,
\(
1 \mathrm{~atm}=1.013 \times 10^5 \mathrm{~Pa}
\)
Other practical units of atmospheric pressure are bar and torr \((\mathrm{mm}\) of Hg\() . \quad 1 \mathrm{~atm}=1.01 \mathrm{bar}=760\) torr
In the experiment it is found that the mercury column in the barometer has a height of about 76 cm at sea level equivalent to one atmosphere ( 1 atm ). A common way of stating pressure is in terms of cm or mm of mercury \((\mathrm{Hg})\). A pressure equivalent of 1 mm is called a torr (after Torricelli).
1 torr \(=133 \mathrm{~Pa}\).
The mm of Hg and torr are used in medicine and physiology. In meteorology, a common unit is the bar and millibar.
\(1 \mathrm{bar}=10^5 \mathrm{~Pa}\)
Example 2: An open container has dimensions of \(4.0 \mathrm{~m} \times 5.0 \mathrm{~m}\) and height of 3.0 m.
(i) Find the weight of the air in the container at \(20^{\circ} \mathrm{C}\).
(ii) What is the weight of an equal volume of water? Also, find pressure at the base of container due to this weight of water.
(iii) What is the total downward force on the base of the container due to air pressure of 1.0 atm ?
(Take, the densities of air and water as \(1.2 \mathrm{~kg} / \mathrm{m}^3\) and \(10^3 \mathrm{~kg} / \mathrm{m}^3\), respectively.)
Solution: It is given that, density of air, \(\rho_{\text {air }}=1.2 \mathrm{~kg} / \mathrm{m}^3\) and density of water, \(\rho_{\text {water }}=10^3 \mathrm{~kg} / \mathrm{m}^3\)
(i) The volume of air in the container
\(
=(4.0)(5.0)(3.0)=60 \mathrm{~m}^3
\)
\(
\begin{aligned}
\therefore \text { The mass of the air is } m_{\text {air }} & =\rho_{\text {air }} \cdot V_{\text {air }} \\
& =(1.2)(60)=72 \mathrm{~kg}
\end{aligned}
\)
The weight of the air is \(w_{\text {air }}=m_{\text {air }} g=(72)(9.8)=705.6 \mathrm{~N}\)
(ii) The weight of water, \(w_{\text {water }}=m_{\text {water }} g=\rho_{\text {water }} \cdot V_{\text {water }} g\)
\(
=\left(10^3\right)(60)(9.8)=5.9 \times 10^5 \mathrm{~N}
\)
Pressure at the base due to this weight,
\(
p=\frac{F(=w)}{A}=\frac{5.9 \times 10^5}{4 \times 5}=2.95 \times 10^4 \mathrm{Nm}^{-2}
\)
(iii) The downward force on the base is
\(
\begin{aligned}
& =(\text { Air pressure }) \text { (Surface area) } \\
& =\left(1.013 \times 10^5\right)(4.0 \times 5.0) \simeq 2.0 \times 10^6 \mathrm{~N}
\end{aligned}
\)
Density of a liquid
Density ( \(\rho\) ) of a liquid or any substance is defined as the mass per unit volume.
\(
\rho=\frac{\text { Mass }}{\text { Volume }}=\frac{m}{V}
\)
Where mass of the fluid is \(m\) occupying volume \(V\).
If the volume is small we can write,
Density, \(\rho=\frac{\text { Mass }}{\text { Volume }}=\lim _{\Delta V \rightarrow 0} \frac{\Delta m}{\Delta V}=\frac{d m}{d V}\)
SI unit of density is \(\mathrm{kgm}^{-3}\) and its dimensions are \(\left[\mathrm{ML}^{-3} \mathrm{~T}^0\right]\) and CGS unit is \(\mathrm{gcm}^{-3}\) with \(1 \mathrm{gcm}^{-3}=10^3 \mathrm{~kg} \mathrm{~m}^{-3}\).
Relative density (RD)
In case of a liquid (or a substance), sometimes an another term relative density (RD) or specific gravity is defined. It is the ratio of density of the liquid (or a substance) to the density of water at \(4^{\circ} \mathrm{C}\).
Hence, \(\quad \mathrm{RD}=\frac{\text { Density of liquid or substance }}{\text { Density of water at } 4^{\circ} \mathrm{C}}\)
Relative density is a pure ratio, so it has no units.
Density of water at \(4^{\circ} \mathrm{C}\) in CGS is \(1 \mathrm{gcm}^{-3}\). Therefore, numerically the relative density and density of a substance in CGS are equal. In SI unit, the density of water at \(4^{\circ} \mathrm{C}\) is \(1000 \mathrm{kgm}^{-3}\).
Example 3: Relative density of an oil is 0.8. Find the absolute density of oil in CGS and SI units.
Solution: To find the absolute density of oil from its relative density, we use the following relationship:
\(
\text { Relative Density }(\mathrm{RD})=\frac{\text { Density of the substance }\left(\rho_s\right)}{\text { Density of water }\left(\rho_w\right) \text { at } 4^{\circ} \mathrm{C}}
\)
Therefore, the absolute density is:
\(
\rho_s=\mathrm{RD} \times \rho_w
\)
In CGS Units:
In the CGS system, the density of water is approximately \(1 \mathrm{~g} / \mathrm{cm}^3\).
\(
\begin{gathered}
\rho_{\mathrm{oil}}=0.8 \times 1 \mathrm{~g} / \mathrm{cm}^3 \\
\rho_{\mathrm{oil}}=0.8 \mathrm{~g} / \mathrm{cm}^3
\end{gathered}
\)
In SI Units:
In the SI system, the density of water is \(1000 \mathrm{~kg} / \mathrm{m}^3\).
\(
\begin{gathered}
\rho_{\text {oil }}=0.8 \times 1000 \mathrm{~kg} / \mathrm{m}^3 \\
\rho_{\text {oil }}=800 \mathrm{~kg} / \mathrm{m}^3
\end{gathered}
\)
Summary:
Absolute density (CGS): \(0.8 \mathrm{~g} / \mathrm{cm}^3\)
Absolute density (SI): \(800 \mathrm{~kg} / \mathrm{m}^3\)
Density of a mixture of two or more liquids
Case I: Suppose two liquids of densities \(\rho_1\) and \(\rho_2\) having masses \(m_1\) and \(m_2\) are mixed together. Then, the density of the mixture will be
\(
\begin{aligned}
\rho & =\frac{\text { Total mass }}{\text { Total volume }} \\
& =\frac{\left(m_1+m_2\right)}{\left(V_1+V_2\right)}=\frac{\left(m_1+m_2\right)}{\left(\frac{m_1}{\rho_1}+\frac{m_2}{\rho_2}\right)}
\end{aligned}
\)
\(
\text { If } m_1=m_2 \text {, then } \rho=\frac{2 \rho_1 \rho_2}{\rho_1+\rho_2} \text {. }
\)
Case II: If you mix equal volumes (for example, 1 liter of oil and 1 liter of water), we can replace \(V_1\) and \(V_2\) with a single variable, \(V\) :
\(
\rho=\frac{\rho_1 V+\rho_2 V}{V+V}
\)
Now, we factor out the \(V\) :
\(
\rho=\frac{V\left(\rho_1+\rho_2\right)}{2 V}
\)
The \(V\) cancels out, leaving you with the formula you provided:
\(
\rho=\frac{\rho_1+\rho_2}{2}
\)
Example 4: Two liquids of densities \(\rho\) and \(3 \rho\) having volumes \(3 V\) andV are mixed together. Find density of the mixture.
Solution: Given, density of first liquid, \(\rho_1=\rho\)
Density of second liquid, \(\rho_2=3 \rho\)
Volume of first liquid, \(V_1=3 \mathrm{~V}\)
Volume of second liquid, \(V_2=V\)
\(\therefore\) Density of the mixture,
\(
\begin{aligned}
\rho_m & =\frac{\rho_1 V_1+\rho_2 V_2}{V_1+V_2} \\
& =\frac{\rho \times 3 V+3 \rho \times V}{3 V+V}=\frac{6 \rho}{4}=\left(\frac{3}{2}\right) \rho
\end{aligned}
\)
Effect of temperature on density
When the temperature of a liquid is increased, the mass remains the same while the volume is increased (Since the molecules move more vigorously at higher temperatures, they push further apart, increasing the total volume while the mass stays constant) and hence, the density of the liquid decreases.
Density (\(\rho\)) and Volume (\(V\)) are inversely proportional for a fixed mass.
\(
\rho \propto \frac{1}{V}
\)
This means if volume goes up (expansion), density must go down.
When you heat a liquid, the new volume (\(V^{\prime}\)) is determined by the coefficient of volume expansion \((\gamma)\) :
\(
V^{\prime}=V(1+\gamma \Delta \theta)
\)
By substituting the volume expansion into the density ratio, we get:
\(
\frac{\rho^{\prime}}{\rho}=\frac{V}{V^{\prime}}=\frac{V}{V(1+\gamma \Delta \theta)}
\)
Canceling out the \(V\) gives us the standard formula for density at a new temperature:
\(
\rho^{\prime}=\frac{\rho}{1+\gamma \Delta \theta}
\)
Important Physics Note: The Binomial Approximation
In many physics problems, \(\gamma\) is a very small number (for example, for water, \(\gamma \approx 2.1 \times 10^{-4} /{ }^{\circ} \mathrm{C}\)).
If the temperature change \(\Delta \theta\) is relatively small, we can use a mathematical shortcut called the Binomial Theorem to rewrite the formula. Since \((1+x)^{-1} \approx 1-x\) when \(x\) is very small, the formula is often written as:
\(
\rho^{\prime} \approx \rho(1-\gamma \Delta \theta)
\)
Exception to the rule: Keep in mind that water is a famous rebel! Between \(0^{\circ} \mathrm{C}\) and \(4^{\circ} \mathrm{C}\), water actually contracts when heated (anomalous expansion). In that specific range, its density actually increases as it warms up.
Effect of pressure on density
This derivation connects the concepts of Fluid Mechanics with Elasticity. It shows that while we often call liquids “incompressible,” they do have a measurable response to extreme pressure, governed by the Bulk Modulus (\(B\)). Just like with temperature, the mass of the fluid stays the same. If you squeeze a fluid (increase pressure), you force the molecules slightly closer together, decreasing the volume (\(V\)) and increasing the density (\(\rho\)).
The Bulk Modulus measures how resistant a substance is to compression. It is defined as:
\(
B=\frac{\text { Stress }}{\text { Strain }}=\frac{-d p}{d V / V}
\)
From this, we can find the change in volume \((d V)\) :
\(
d V=-\frac{V \cdot d p}{B}
\)
(The negative sign indicates that as pressure \(d p\) increases, volume \(V\) decreases).
By substituting this change into the ratio of densities, we get:
\(
\frac{\rho^{\prime}}{\rho}=\frac{V}{V+d V}=\frac{V}{V-\frac{V \cdot d p}{B}}
\)
By factoring out and canceling \(V\) :
\(
\frac{\rho^{\prime}}{\rho}=\frac{1}{1-\frac{d p}{B}}
\)
Rearranging gives you the final formula for the new density:
\(
\rho^{\prime}=\frac{\rho}{1-\frac{d p}{B}}
\)
Here, \(d p=\) change in pressure
and \(B=\) bulk modulus of elasticity of the liquid.
Example 5: The thermal coefficient of volume expansion of a liquid is \(5 \times 10^{-4} \mathrm{~K}^{-1}\). If its temperature is increased by \(30^{\circ} \mathrm{C}\), find the ratio of new density to the previous one.
Solution: Given, thermal coefficient of volume expansion,
\(
\gamma=5 \times 10^{-4} \mathrm{~K}^{-1}
\)
Rise in temperature, \(\Delta \theta=30^{\circ} \mathrm{C}\)
\(
\therefore \quad \quad \rho^{\prime}=\frac{\rho}{1+\gamma \Delta \theta}
\)
\(
\begin{aligned}
\frac{\rho^{\prime}}{\rho} & =\frac{1}{1+\gamma \Delta \theta} \\
& =\frac{1}{1+\left(5 \times 10^{-4}\right)(30)} \simeq 0.98
\end{aligned}
\)
Example 6: The bulk modulus of a liquid is \(8 \times 10^9 \mathrm{Nm}^{-2}\) and its density is \(11 \mathrm{~g} \mathrm{~cm}^{-3}\). What will be the density of liquid under a pressure of \(20,000 \mathrm{Ncm}^{-2}\) ?
Solution: Given, bulk modulus of liquid, \(B=8 \times 10^9 \mathrm{Nm}^{-2}\) Density of liquid, \(\rho=11 \mathrm{~g} \mathrm{~cm}^{-3}\) and pressure, \(p=20000 \mathrm{Ncm}^{-2}\)
\(\therefore\) New density of liquid, \(\rho^{\prime}=\frac{\rho}{\left(1-\frac{d p}{B}\right)}\) or \(\frac{\rho}{\left(1-\frac{p}{B}\right)}\)
\(
\begin{aligned}
& =\frac{11}{1-\frac{20000}{8 \times 10^9 \times 10^{-4}}} \\
& =\frac{11 \times 40}{39}=\frac{440}{39} \mathrm{gcm}^{-3}
\end{aligned}
\)
Pressure due to liquid column
A tall beaker is filled with liquid so that it forms a liquid column (a liquid column is a vertical, gravity-supported column of liquid within a container or tube, commonly used to measure pressure based on the height of the fluid). The area of cross section at the bottom is \(A\). The density of the liquid is \(\rho\). The height of the liquid column is \(h\). In other words the depth of the water from the top level surface is ‘ \(h\) ‘ as shown in Figure below.

We know that thrust at the bottom of the column \(({F})=\) weight of the liquid.
Therefore, \({F}={mg} \dots(1)\)
We can get the mass of the liquid by multiplying the volume of the liquid and its density.
Mass, \({m}=\rho {V} \dots(2)\)
Volume of the liquid column, \({V}=\) Area of cross section \(({A}) \times\) Height \(({h})={Ah} \dots(3)\)
Substituting (3) in (2)
Hence, mass, \(m=\rho A h \dots(4)\)
Substituting (4) in (1)
Force \(F=m g=\rho A h g\)
Pressure, \(P=\frac{\text { Thrust }({F})}{\operatorname{Area}({A})}=\frac{{mg}}{{A}}=\frac{\rho({Ah}) {g}}{{A}}=\rho {hg}\)
\(\therefore\) Pressure due to a liquid column, \({P}={h} \rho {g}\)
This expression shows that pressure in a liquid column is determined by depth, density of the liquid and the acceleration due to gravity. Pressure exerted by a liquid at a point is determined by,
- depth (\(h\))
- density of the liquid ( \(\rho\))
- acceleration due to gravity (\(g\)).
Interestingly, the final expression for pressure does not have the term area \(A\) in it. Thus pressure at a given depth does not depend upon the shape of the vessel containing the liquid or the amount of liquid in the vessel. It only depends on the depth. In Figure below, the pressure is the same even though the containers have different amounts of liquid in them, and are of different shapes.

Variation of fluid pressure with depth
Consider a fluid of density \(\rho\) is kept at rest, in a cylindrical vessel of height \(h\) as shown in figure.

In Figure above point 1 is at height \(h\) above a point 2. The pressures at points 1 and 2 are \(p_1\) and \(p_2\) respectively. Consider a cylindrical element of fluid having area of base \(A\) and height \(h\). In static equilibrium, a fluid is at rest, meaning it has zero acceleration. According to Newton’s Second Law (\(\Sigma F=m a\)), if acceleration (\(a\)) is zero, the net force (\(\Sigma F\)) acting on the fluid must also be zero. Therefore, all vertical forces (like pressure and gravity) must balance to zero, or the fluid would accelerate.
Identifying the Vertical Forces:
There are three main forces acting on your cylinder:
Force at the Top: The pressure \(p_1\) pushes down on the top area \(A\).
\(F_{\text {top }}=p_1 A\) (Downward)
Force at the Bottom: The pressure \(p_2\) pushes up on the bottom area \(\boldsymbol{A}\).
\(F_{\text {bottom }}=p_2 A\) (Upward)
Weight of the Fluid: The Earth pulls the mass of the fluid inside the cylinder downward.
\(W=m g\) (Downward)
Because the fluid isn’t moving up or down, the Upward Forces must equal the Downward
Forces:
\(
\begin{aligned}
& \text { Upward Forces }=\text { Downward Forces } \\
& \qquad p_2 A=p_1 A+m g
\end{aligned}
\)
Substituting Mass for Density:
To make this useful for any fluid, we replace mass (\(m\)) with the fluid’s density (\(\rho\)) and volume ( \(V)\).
We know that \(m=\rho V\)
The volume of a cylinder is Area × Height, so \(V=A \cdot h\)
Therefore, \(m g=(\rho \cdot A \cdot h) g\)
Now, substitute this back into our force equation:
\(
p_2 A=p_1 A+\rho A h g
\)
\(
p_2=p_1+\rho g h
\)
\(
p_2-p_1=\rho g h \dots(i)
\)
This is why the pressure at the bottom is higher. The pressure at point 2 is simply the pressure at point 1 plus the weight of the fluid column pressing down per unit area.
From the above equation we can see the Pressure difference depends on the vertical distance \(h\) between the points ( 1 and 2), mass density of the fluid \(\rho\) and acceleration due to gravity \(g\).
If the point 1 under discussion is shifted to the top of the fluid (say, water), which is open to the atmosphere, \({p}_1\) may be replaced by atmospheric pressure \(\left({p}_{{a}}\right)\) and we replace \({p}_2\) by \(p\). Then Eq. (i) gives
\(
p=p_{\mathrm{a}}+\rho g h \dots(ii)
\)
Thus, the pressure \(p\), at depth below the surface of a liquid open to the atmosphere is greater than atmospheric pressure by an amount \(\rho g h\). The excess of pressure, \(p-p_{{a}}\), at depth \(h\) is called a gauge pressure at that point.
According to Eq. (ii), pressure increases linearly with depth, if \(\rho\) and \(g\) are uniform.
A graph between \(p\) and \(h\) is shown below

The area of the cylinder is not appearing in the expression of absolute pressure in Eq. (ii). Thus, the height of the fluid column is important and not cross-sectional or base area or the shape of the container. The liquid pressure is the same at all points at the same horizontal level (same depth).
Absolute pressure and gauge pressure
The excess pressure above atmospheric pressure is usually called gauge pressure and the total pressure is called absolute pressure. Thus,
Gauge pressure = Absolute pressure – Atmospheric pressure
Absolute pressure is always greater than or equal to zero. While gauge pressure can be negative also.
Example 7: Water is filled in a flask up to a height of 20 cm. The bottom of the flask is circular with radius 10 cm. If the atmospheric pressure is \(1.01 \times 10^5 \mathrm{~Pa}\), find the force exerted by the water on the bottom. Take \(g=10 \mathrm{~m} \mathrm{~s}^{-2}\) and density of water \(=1000 \mathrm{~kg} \mathrm{~m}^{-3}\).
Solution: The pressure at the surface of the water is equal to the atmospheric pressure \(P_0\). The pressure at the bottom is
\(
\begin{aligned}
&\begin{aligned}
P & =P_0+h \rho g \\
& =1.01 \times 10^5 \mathrm{~Pa}+(0.20 \mathrm{~m})\left(1000 \mathrm{~kg} \mathrm{~m}^{-3}\right)\left(10 \mathrm{~m} \mathrm{~s}^{-2}\right) \\
& =1.01 \times 10^5 \mathrm{~Pa}+0.02 \times 10^5 \mathrm{~Pa} \\
& =1.03 \times 10^5 \mathrm{~Pa}
\end{aligned}\\
&\begin{aligned}
\text { The area of the bottom }=\pi r^2 & =3.14 \times(0.1 \mathrm{~m})^2 \\
& =0.0314 \mathrm{~m}^2
\end{aligned}\\
&\text { The force on the bottom is, therefore, }\\
&\begin{aligned}
F & =P \pi r^2 \\
=\left(1.03 \times 10^5 \mathrm{~Pa}\right) \times\left(0.0314 \mathrm{~m}^2\right) & =3230 \mathrm{~N} .
\end{aligned}
\end{aligned}
\)
Example 8: The density of the atmosphere at sea level is \(1.29 \mathrm{~kg} / \mathrm{m}^3\). Assume that it does not change with altitude. Then how high would the atmosphere extend?
Solution: To determine how high the atmosphere would extend under the assumption of constant density, we can use the relationship between pressure, density, and height for a fluid column. We know, \(P=P_{\mathrm{0}}+\rho g h\)
Thus, the pressure \(P\), at depth below the surface of a liquid open to the atmosphere is greater than atmospheric pressure by an amount \(\rho g h\).
Identify Given Information and Constants:
Density of the atmosphere \((\rho): 1.29 \mathrm{~kg} / \mathrm{m}^3\) (given).
Pressure at sea level (\(P_0\)): Standard atmospheric pressure is approximately \(1.01325 \times 10^5 \mathrm{~Pa}\) (or \(\mathrm{N} / \mathrm{m}^2\)).
Acceleration due to gravity (\(g\)): Approximately \(9.81 \mathrm{~m} / \mathrm{s}^2\).
The Formula:
For a fluid of constant density, the pressure \(P\) at the bottom of a column of height \(h\) is given by:
\(
P_0=\rho g h
\)
To find the height \(h\), we rearrange the formula:
\(
h=\frac{P_0}{\rho g}=\frac{101,325 \mathrm{~Pa}}{1.29 \mathrm{~kg} / \mathrm{m}^3 \times 9.81 \mathrm{~m} / \mathrm{s}^2}=\approx 8 \mathrm{~km}
\)
If the atmosphere had a uniform density of \(1.29 \mathrm{~kg} / \mathrm{m}^3\) throughout, it would extend to a height of approximately 8,000 meters (or about 8 km).
Example 9: At a depth of 1000 m in an ocean (a) what is the absolute pressure? (b) What is the gauge pressure? (c) Find the force acting on the window of area \(20 \mathrm{~cm} \times 20 \mathrm{~cm}\) of a submarine at this depth, the interior of which is maintained at sealevel atmospheric pressure. (The density of sea water is \(1.03 \times 10^3 \mathrm{~kg} \mathrm{~m}^{-3}\), \(g=10 \mathrm{~m} \mathrm{~s}^{-2}\).)
Solution: Given Data:
Depth (\(h\)): 1000 m
Density of sea water \((\rho)\) : \(1.03 \times 10^3 \mathrm{~kg} \mathrm{~m}^{-3}\)
Acceleration due to gravity ( \(g\)): \(10 \mathrm{~m} \mathrm{~s}^{-2}\)
Atmospheric pressure \(\left(P_a\right): 1.01 \times 10^5 \mathrm{~Pa}\)
Window Area (A): \(20 \mathrm{~cm} \times 20 \mathrm{~cm}=0.2 \mathrm{~m} \times 0.2 \mathrm{~m}=0.04 \mathrm{~m}^2\)
(a) Absolute Pressure
The absolute pressure \((P)\) at a depth \(h\) is the sum of the atmospheric pressure and the pressure exerted by the water column (gauge pressure).
\(
P=P_a+\rho g h
\)
Substituting the values:
\(
\begin{gathered}
P=\left(1.01 \times 10^5 \mathrm{~Pa}\right)+\left(1.03 \times 10^3 \mathrm{~kg} \mathrm{~m}^{-3} \times 10 \mathrm{~m} \mathrm{~s}^{-2} \times 1000 \mathrm{~m}\right) \\
P=1.01 \times 10^5 \mathrm{~Pa}+103 \times 10^5 \mathrm{~Pa} \\
P=104.01 \times 10^5 \mathrm{~Pa} \approx 1.04 \times 10^7 \mathrm{~Pa}(\text { or } 104 \mathrm{~atm})
\end{gathered}
\)
(b) Gauge Pressure
Gauge pressure (\(P_g\)) is the difference between the absolute pressure and the atmospheric pressure.
\(
\begin{gathered}
P_g=P-P_a=\rho g h \\
P_g=1.03 \times 10^3 \times 10 \times 1000 \\
P_g=1.03 \times 10^7 \mathrm{~Pa}(\text { or } \approx 103 \mathrm{~atm})
\end{gathered}
\)
(c) Force acting on the window
The interior of the submarine is maintained at sea-level atmospheric pressure (\(P_a\)). Therefore, the net pressure acting on the window is the difference between the external absolute pressure and the internal atmospheric pressure, which is equal to the gauge pressure.
Net Pressure \((\triangle P)=P-P_a=P_g\)
The force \((F)\) is calculated as:
\(
\begin{gathered}
F=\Delta P \times A \\
F=\left(1.03 \times 10^7 \mathrm{~Pa}\right) \times\left(0.04 \mathrm{~m}^2\right) \\
F=4.12 \times 10^5 \mathrm{~N}
\end{gathered}
\)
Hydrostatic paradox
Consider three vessels A, B and C [Figure below] of different shapes. They are connected at the bottom by a horizontal pipe. On filling with water, the level in the three vessels is the same, though they hold different amounts of water. This is so because water at the bottom has the same pressure below each section of the vessel.

Example 10: What is the pressure on a swimmer 10 m below the surface of a lake?
Solution: Here
\(h=10 \mathrm{~m}\) and \(\rho=1000 \mathrm{~kg} \mathrm{~m}^{-3}\). Take \(\mathrm{g}=10 \mathrm{~m} \mathrm{~s}^{-2}\)
From Eq. (ii)
\(
\begin{aligned}
p & =p_{\mathrm{a}}+\rho g h \\
& =1.01 \times 10^5 \mathrm{~Pa}+1000 \mathrm{~kg} \mathrm{~m}^{-3} \times 10 \mathrm{~m} \mathrm{~s}^{-2} \times 10 \mathrm{~m} \\
& =2.01 \times 10^5 \mathrm{~Pa} \\
& \approx 2 \mathrm{~atm}
\end{aligned}
\)
This is a \(100 \%\) increase in pressure from surface level. At a depth of 1 km , the increase in pressure is 100 atm ! Submarines are designed to withstand such enormous pressures.
Important points related to fluid pressure
Important points related to fluid pressure are given below:
- At a point in the liquid column, the pressure applied on it is same in all directions: This is known as Pascal’s Law. In a fluid, the molecules are in constant, random motion and are free to slide past one another. If pressure were higher in one direction than another, the fluid would simply flow in the direction of lower pressure until the forces balanced out. Because a fluid at rest isn’t moving, the “push” must be equal from the top, bottom, and sides.
- In a liquid, pressure will be same at all points at the same level (height): From the formula we derived earlier \(\left(p_2=p_1+\rho g h\right)\), you can see that pressure only changes if the height (\(h\)) changes.
Consider two points, A and B , at the same horizontal level. If the pressure at A were higher than at B, there would be a net horizontal force pushing the liquid from A toward B. Since the liquid is “at rest” (static), there can be no net force. Therefore, the pressure must be identical across any horizontal plane. - The pressure exerted by a liquid depends only on the height of fluid column and is independent of the shape of the containing vessel: In the formula \(p=\rho g h\), there is no variable for “Width,” “Volume,” or “Shape.”

- Consider following shapes of vessels: Pressure at the base of each vessel,
\(
\begin{aligned}
& p_x=p_y=p_z=p_0+\rho g h \text { but weight, } w_x \neq w_y \neq w_z \\
& \text { where, } \rho=\text { density of liquid in each vessel, } \\
& \qquad h=\text { height of liquid in each vessel }
\end{aligned}
\)
and \(\quad p_0=\) atmospheric pressure.

- In the figure, a block of mass \(m\) floats over a fluid surface, If \(\rho=\) density of the liquid,
\(A=\) area of the block, then
Pressure at the base of the vessel is
\(
p=p_0+\rho g h+\frac{m g}{A}
\)

Example 11: Find the pressure exerted below a column of water, open to the atmosphere, at depth 10 m (Take, density of water \(=1 \times 10^3 \mathrm{kgm}^{-3}, g=10 \mathrm{~ms}^{-2}\) ).
Solution: Pressure at depth of 10 m ,
\(
\begin{aligned}
p & =p_a+\rho g h \\
& =1.013 \times 10^5 \mathrm{~Pa}+\left(1 \times 10^3 \mathrm{kgm}^{-3}\right)\left(10 \mathrm{~ms}^{-2}\right)(10 \mathrm{~m}) \\
& =1.013 \times 10^5 \mathrm{~Pa}+1 \times 10^5 \mathrm{~Pa} \\
& =2.013 \times 10^5 \mathrm{~Pa} \\
& \approx 2 \mathrm{~atm}
\end{aligned}
\)
Example 12: For the arrangement shown in the figure, what is the density of oil?

Solution: The fundamental rule for a fluid at rest is that pressure is the same at all points at the same horizontal level within the same continuous liquid. In the diagram, the dashed line represents this interface level.
To maintain equilibrium, the pressure exerted by the column of oil on the left must equal the pressure exerted by the column of water on the right at that dashed line.
Identifying the Variables:
From the figure, we can identify the heights of the two liquid columns above the reference line:
Height of Water (\(l\)): 135 mm
Height of Oil (\(h_{\text {oil }}\)): This is the sum of the water height and the extra height \(d\).
\(h_{\text {oil }}=l+d=135 \mathrm{~mm}+12.3 \mathrm{~mm}=147.3 \mathrm{~mm}\)
Density of Water \(\left(\rho_w\right): 1000 \mathrm{~kg} / \mathrm{m}^3\) (standard value)
Setting Up the Equation:
The pressure at the interface level in both arms of the U-tube is equal: (the reason pressure is the same at the interface level (the dashed line in your diagram) boils down to the fact that the fluid is in static equilibrium.)
\(
P_{\text {left }}=P_{\text {right }}
\)
\(
p_0+\rho_{o i l} g (l+d)=p_0+\rho_w g l
\)
By canceling atmospheric pressure ( \(p_0\) ) and gravity (\(g\)) from both sides, the equation simplifies to:
\(
\rho_{o i l} \cdot(l+d)=\rho_w \cdot l
\)
Final Calculation:
Now, we rearrange the formula to solve for the density of oil (\(\rho_{\text {oil }}\)):
\(
\begin{gathered}
\rho_{\text {oil }}=\frac{\rho_w \cdot l}{l+d} \\
\rho_{\text {oil }}=\frac{1000 \mathrm{~kg} / \mathrm{m}^3 \cdot 135 \mathrm{~mm}}{135 \mathrm{~mm}+12.3 \mathrm{~mm}} \\
\rho_{\text {oil }}=\frac{135,000}{147.3} \approx 916.5 \mathrm{~kg} / \mathrm{m}^3
\end{gathered}
\)
Summary of Result: The density of the oil is approximately \(916 \mathrm{~kg} / \mathrm{m}^3\). Since the oil column is taller than the water column to exert the same pressure, it makes sense that the oil is less dense than water.
Pascal’s law
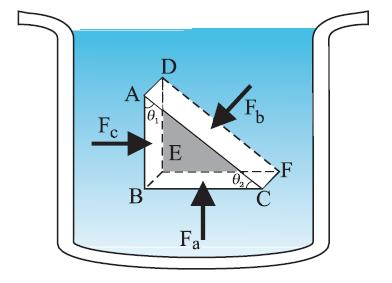
The French scientist Blaise Pascal observed that the pressure in a fluid at rest is the same at all points if they are at the same height. Figure above shows an element in the interior of a fluid at rest. This element \(\mathrm{ABC}-\mathrm{DEF}\) is in the form of a right-angled prism. In principle, this prismatic element is very small so that every part of it can be considered at the same depth (same height) from the liquid surface and therefore, the effect of the gravity is the same at all these points. But for clarity we have enlarged this element. The forces on this element are those exerted by the rest of the fluid and they must be normal to the surfaces of the element as discussed above. Thus, the fluid exerts pressures \(P_{\mathrm{a}}, P_{\mathrm{b}}\) and \(P_{\mathrm{c}}\) on this element of area corresponding to the normal forces \(F_{\mathrm{a}}, F_{\mathrm{b}}\) and \(F_{\mathrm{c}}\) as shown in the figure on the faces \(\mathrm{BEFC, ADFC}\) and \(\mathrm{ADEB}\) denoted by \(A_{\mathrm{a}}, A_{\mathrm{b}}\) and \(A_c\) respectively. Then
\(
\frac{F_b}{A_b}=\frac{F_c}{A_c}=\frac{F_a}{A_a} ; \quad P_b=P_c=P_a
\)
Hence, pressure exerted is same in all directions in a fluid at rest.
Pascal’s Law Derivation:
Let us consider a right-angled triangle(with sides p, q, and r) prism (height s) submerged in the liquid of density \(\rho\), also assume the size of the submersed element is negligible with compare to the volume of the liquid, and all the points on the element experience the same gravitational force.

Now, the area of the faces PQRS, PSUT, and QRUT of the prism is ps, qs, and rs respectively. Also, assume the pressure applied by the liquid on these faces is \(\mathrm{P}_1\), \(P_2\), and \(P_3\) respectively.
Exerted force by this pressure to the faces in the perpendicular inward direction is \(F_1, F_2\), and \(F_3\).
Thus, \(F_1=P_1 \times\) Area of PQRS \(=P_1 \times p s\)
\(F_2=P_2 \times\) Area of PSUT \(=P_2 \times q s\)
\(F_3=P_3 \times\) Area of QRUT \(=P_3 \times r s\)
Now, in triangle PQT,
\(\sin \theta=p / r\) and \(\cos \theta=q / r\)
The net force on the prism will be zero since the prism is in equilibrium.
\(F_3 \sin \theta=F 1\) and \(F_3 \cos \theta=F_2\) (putting values of \(F_1, F_2\), and \(F_3\) from the above values)
\(
\begin{aligned}
& \Rightarrow P_3 \times r s \times p / r=P_1 \times p s \text { and } P_3 \times r s \times q / r=P_2 \times q s \\
& \Rightarrow P_3=P_1 \text { and } P_3=P_2
\end{aligned}
\)
Thus, \(\mathrm{P}_1=\mathrm{P}_2=\mathrm{P}_3\)
Therefore, pressure throughout the liquid remains the same.
Hydraulic lift
A well known application of Pascal’s law is the hydraulic lift used to support or lift heavy objects. A number of devices, such as hydraulic lift and hydraulic brakes, are based on the Pascal’s law. In these devices, fluids are used for transmitting pressure. It is schematically illustrated in figure.

In a hydraulic lift, as shown in Figure above, two pistons are separated by the space filled with a liquid. A piston of small cross-section \(A_1\) is used to exert a force \(F_1\) directly on the liquid. The pressure \(P=\frac{F_1}{A_1}\) is transmitted throughout the liquid to the larger cylinder attached with a larger piston of area \(A_2\), which results in an upward force of \(P \times A_2\). Therefore, the piston is capable of supporting a large force (large weight of, say a car, or a truck, placed on the platform)
\(F_2=P A_2=\frac{F_1 A_2}{A_1}\).
By changing the force at \(A_1\), the platform can be moved up or down. Thus, the applied force has been increased by a factor of \(\frac{A_2}{A_1}\) and this factor is the mechanical advantage of the device.
Now, since \(A_2 \gg A_1\), therefore \(F_2 \gg F_1\). Thus, hydraulic lift is a force multiplying device with a multiplication factor equal to the ratio of the areas of the two pistons. Dentist’s chairs, car lifts and jacks, hydraulic elevators and hydraulic brakes all use this principle.
Example 13: Two pistons of a hydraulic machine have diameters 20 cm and 2 cm . Find the force exerted on the larger piston when 50 kg -wt is placed on the smaller piston. When the smaller piston moves in through 5 cm , by what distance, the other piston moves out?
Solution: For smaller piston, area, \(A_1=\pi \times(1)^2\)
For larger piston, area, \(A_2=\pi \times(10)^2\)
\(\therefore\) Force exerted on the larger piston,
\(
\begin{aligned}
F_2 & =\frac{A_2}{A_1} \times F_1 \\
& =\frac{\pi(10)^2}{\pi(1)^2} \times 50 \times 9.8 \\
& =100 \times 50 \times 9.8 \\
& \simeq 5 \times 10^4 \mathrm{~N}
\end{aligned}
\)
This is the force exerted on the larger piston. The liquids are considered incompressible. Therefore, volume covered by movement of smaller piston inwards equal to the outward movement of larger piston.
\(
\begin{aligned}
\therefore \quad L_1 A_1 & =L_2 A_2 \\
\Rightarrow \quad L_2 & =\left(\frac{A_1}{A_2}\right) L_1 \\
& =\frac{\pi(1 \mathrm{~cm})^2}{\pi(10 \mathrm{~cm})^2} \times 5 \mathrm{~cm} \\
& =\frac{1}{100} \times 5 \mathrm{~cm} \\
& =0.05 \mathrm{~cm}
\end{aligned}
\)
So, the distance moved out by the larger piston is 0.05 cm.
Example 14: In a car lift compressed air exerts a force \(F_1\) on a small piston having a radius of 5.0 cm . This pressure is transmitted to a second piston of radius 15 cm (Fig 9.7). If the mass of the car to be lifted is 1350 kg , calculate \(F_1\). What is the pressure necessary to accomplish this task? \(\left(g=9.8 \mathrm{~ms}^{-2}\right)\).
Solution: Since pressure is transmitted undiminished throughout the fluid,
\(
\begin{aligned}
F_1=\frac{A_1}{A_2} F_2= & \frac{\pi\left(5 \times 10^{-2} \mathrm{~m}\right)^2}{\pi\left(15 \times 10^{-2} \mathrm{~m}\right)^2}\left(1350 \mathrm{~kg} \times 9.8 \mathrm{~m} \mathrm{~s}^{-2}\right) \\
& =1470 \mathrm{~N} \\
& \approx 1.5 \times 10^3 \mathrm{~N}
\end{aligned}
\)
The air pressure that will produce this force is
\(
P=\frac{F_1}{A_1}=\frac{1.5 \times 10^3 \mathrm{~N}}{\pi\left(5 \times 10^{-2}\right)^2 \mathrm{~m}}=1.9 \times 10^5 \mathrm{~Pa}
\)
This is almost double the atmospheric pressure.
Example 15: Two syringes of different cross-sections (without needles) filled with water are connected with a tightly fitted rubber tube filled with water. Diameters of the smaller piston and larger piston are 1.0 cm and 3.0 cm respectively. (a) Find the force exerted on the larger piston when a force of 10 N is applied to the smaller piston. (b) If the smaller piston is pushed in through 6.0 cm , how much does the larger piston move out?
Solution: (a) Since pressure is transmitted undiminished throughout the fluid,
\(
\begin{aligned}
F_2=\frac{A_2}{A_1} F_1 & =\frac{\pi\left(3 / 2 \times 10^{-2} \mathrm{~m}\right)^2}{\pi\left(1 / 2 \times 10^{-2} \mathrm{~m}\right)^2} \times 10 \mathrm{~N} \\
& =90 \mathrm{~N}
\end{aligned}
\)
(b) Water is considered to be perfectly incompressible. Volume covered by the movement of smaller piston inwards is equal to volume moved outwards due to the larger piston.
\(
\begin{aligned}
& L_1 A_1=L_2 A_2 \\
& \begin{aligned}
L_2=\frac{A_1}{A_2} & L_1=\frac{\pi\left(1 / 2 \times 10^{-2} \mathrm{~m}\right)^2}{\pi\left(3 / 2 \times 10^{-2} \mathrm{~m}\right)^2} \times 6 \times 10^{-2} \mathrm{~m} \\
& \simeq 0.67 \times 10^{-2} \mathrm{~m}=0.67 \mathrm{~cm}
\end{aligned}
\end{aligned}
\)
Note, atmospheric pressure is common to both pistons and has been ignored.
Example 16: Figure shows a hydraulic press with the larger piston of diameter 35 cm at a height of 1.5 m relative to the smaller piston of diameter 10 cm. The mass on the smaller piston is 20 kg. What is the force exerted on the load by the larger piston? The density of oil in the press is \(750 \mathrm{kgm}^{-3}\). (Take, \(g=9.8 \mathrm{~ms}^{-2}\))
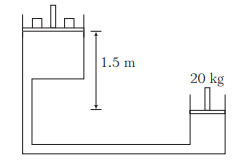
Solution: Pressure on the smaller piston \(=\frac{20 \times 9.8}{\pi \times\left(5 \times 10^{-2}\right)^2} \mathrm{Nm}^{-2}\) Pressure on the larger piston \(=\frac{F}{\pi \times\left(17.5 \times 10^{-2}\right)^2} \mathrm{Nm}^{-2}\) The difference between the two pressures \(=h \rho g\) where,
\(
h=1.5 \mathrm{~m}
\)
and \(\rho=750 \mathrm{kgm}^{-3}\)
Thus, \(1.5 \times 750 \times 9.8=\frac{20 \times 9.8}{\pi \times\left(5 \times 10^{-2}\right)^2}-\frac{F}{\pi \times\left(17.5 \times 10^{-2}\right)^2}\)
which gives, \(\quad F=1.3 \times 10^3 \mathrm{~N}\)
Note Atmospheric pressure is common to both pistons and has been ignored.
Measurement of pressure
Pressure can be measured by using following two devices:
Barometer:
It is a device used to measure atmospheric pressure. In principle, any liquid can be used to fill the barometer, but mercury is the substance of choice because its high density makes possible an instrument of reasonable size. A barometer is an inverted evacuated tube, put over a mercury volume. Outside pressure pushes mercury into tube till the weight of liquid column equalises the force due to external pressure.

In given diagram, in equilibrium,
\(p_1=p_2\)
\(
\begin{array}{ll}
\text { Here, } & p_1=\text { atmospheric pressure }\left(p_0\right) [\text { same as } \left(p_a\right)\\
\text { and } & p_2=0+\rho g h=\rho g h
\end{array}
\)
where, \(\rho=\) density of mercury
\(
\therefore \quad p_0=\rho g h
\)
Thus, the mercury barometer reads the atmospheric pressure \(\left(p_0\right)\) directly from the height of the mercury column.
e.g. If the height of mercury in a barometer is 760 mm , then atmospheric pressure will be
\(
\begin{aligned}
p_0 & =\rho g h \\
& =\left(13.6 \times 10^3\right)(9.8)(0.760) \\
& =1.01 \times 10^5 \mathrm{Nm}^{-2}
\end{aligned}
\)
Manometer:
It is a device used to measure the pressure of a gas inside a container.

The U-shaped tube often contains mercury. Let 1 and 2 are points on same horizontal level, then
\(
p_1=p_2
\)
Here, \(\quad p_1=\) pressure of the gas in the container \((p)\)
and \(\quad p_2=\) atmospheric pressure \(\left(p_0\right)+\rho g h\)
\(
\therefore \quad p=p_0+\rho g h
\)
This can also be written as
\(
p-p_0=\text { gauge pressure }=\rho g h
\)
Here, \(\rho\) is the density of the liquid used in U-tube. Thus, by measuring \(h\), we can find absolute (or gauge) pressure in the vessel.
Example 17: What will be the length of mercury column in a barometer tube, when the atmospheric pressure is 76 cm of mercury and the tube is inclined at an angle of \(30^{\circ}\) with the horizontal direction?
Solution: Here, \(h=76 \mathrm{~cm}, \theta=30^{\circ}\)

If \(l\) is the length of mercury column in a barometer tube, then
\(
\frac{h}{l}=\sin 30^{\circ}
\)
\(
\begin{array}{ll}
\Rightarrow & \frac{76 \mathrm{~cm}}{l}=\frac{1}{2} \\
\Rightarrow & l=2 \times 76=152 \mathrm{~cm}
\end{array}
\)
Example 18: A manometer tube contains a liquid of density \(3 \times 10^3 \mathrm{~kg} \mathrm{~m}^{-3}\). When connected to a vessel containing a gas, the liquid level in the other arm of the tube is higher by 20 cm . When connected to another sample of enclosed gas, the liquid level in the other arm of the manometer tube falls 8 cm below the liquid level in the first arm. Which of the two samples exerts more pressure and by what amount?
Solution:

For Sample 1:
Difference in level of liquids, \(h_1=20 \mathrm{~cm}=0.2 \mathrm{~m}\)
Pressure of the gas in the left arm, \(p_1=p_a+\rho g h_1 \dots(i)\)
For sample 2:

In this case, level of the liquid in the left arm is higher than that in the right arm by 8 cm .
\(\therefore\) Atmospheric pressure \(p_a\) is greater than the pressure exerted by the sample, i.e.
\(
p_a=p_2+\rho g h_2 \Rightarrow p_2=p_a-\rho g h_2 \dots(ii)
\)
Comparing Eqs. (i) and (ii), it is clear that \(p_1>p_2\).
Therefore, the gas in sample 1 exerts greater pressure than that in sample 2.
The difference in the two pressures is
\(
\begin{aligned}
p_1-p_2 & =\left(p_a+\rho g h_1\right)-\left(p_a-\rho g h_2\right) \\
& =\rho g\left(h_1+h_2\right)=\rho g(28 \mathrm{~cm}) \\
& =\left(3 \times 10^3 \mathrm{~kg} \mathrm{~m}^{-3}\right) \times\left(9.8 \mathrm{~ms}^{-2}\right)(0.28 \mathrm{~m}) \\
& =8.23 \times 10^3 \mathrm{~Pa} \approx 8 \mathrm{kPa}
\end{aligned}
\)
Archimedes’ principle
When a body is partially or fully dipped into a fluid, the fluid exerts forces on the body. At any small portion of the surface of the body, the force by the fluid is perpendicular to the surface and is equal to the pressure at that point multiplied by the area (figure below). The resultant of all these contact forces is called the force of buoyancy or buoyant force.
Archimedes’ principle states that when a body is partially or fully dipped into a fluid at rest, the fluid exerts an upward force of buoyancy equal to the weight of the displaced fluid.
Thus, the magnitude of buoyant force \((F)\) which is also called upthrust is given by
\(
F=V \rho_w g
\)
where, \(\quad V=\) immersed volume of solid,
\(\rho_w=\) density of water
and \(\quad g=\) acceleration due to gravity.
The upthrust act vertically upwards through the centre of gravity of displaced fluid.
Archimedes’s principle refers to the upward force of buoyancy that results when a body is submerged in a fluid, whether partially or wholly. We will find that the main reason for buoyancy is the difference in pressure at different depths. Due to buoyancy, an object appear lighter when submerged. The difference of actual weight of a body and the buoyancy force is called the apparent weight of the body.
Block fully submerged in water
Let’s look at a rectangular block fully submerged in water as in figure below. The pressure from the sides exert forces on the block – they cancel each other out. But, since the pressure at the top surface is less than the pressure at the bottom surface, the forces from those pressures will have a left over upward pointing net force. This is the buoyancy force.
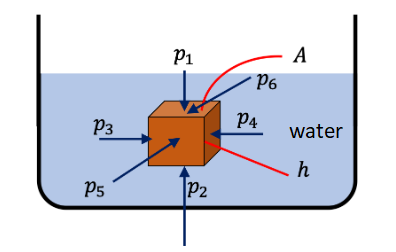
The buoyancy force (\(F_b\)) on the block occurs due to pressure increasing with depth. The net upward force is the buoyancy force, whose magnitude would be \(F_b=p_2 A-p_1 A=\left(p_2-p_1\right) A\). The horizontal forces, \(p_3 A, p_4 A, p_5 A, p_6 A\) are balanced – they are not involved in buoyancy.
The buoyancy force is pointed up and has the magnitude
\(
F_b=p_2 A-p_1 A=\left(p_2-p_1\right) A .
\)
Writing the pressure at a point in the fluid as
\(
p=p_0+\rho g h,
\)
where \(p_0\) is the atmospheric pressure, we can see that
\(
p_2-p_1=\rho_w g h .
\)
where \(\rho_w\) is the density of water and \(h\) the height of the block. Therefore, we can write the force of buoyancy as
\(
F_b=\left(p_2-p_1\right) A=\rho_w g h A=\rho_w g V,
\)
where \(V\) is the volume of the block. Since the block is fully submerged, \(V\) is also the volume of the fluid that was displaced by the block. We see that \(\rho_w V\) is the mass of the water corresponding to the volume of water that was displaced by the submerged block. Therefore, we write the buoyancy force alternately as (in vector form)
\(
\vec{F}_b=\text { Weight of displaced fluid, pointed up. }
\)
This is the statement of the Archimedes’ principle.
Block Partially submerged in water
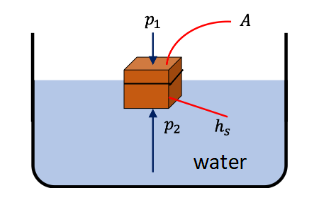
This works also for the partially submerged block. When the block is partially submerged, the volume will not be the volume of the entire block, but of only the part that is actually submerged.
Figure above shows a partially submerged block of full height \(h\), of which only the height \(h_s\) is submerged. Forces from the sides again cancel out, and the upward buiyancy force is
\(
\begin{aligned}
F_b & =\left(p_2-p_1\right) A=\rho_w g h_s A \\
& =W_{\text {displaced fluid }}
\end{aligned}
\)
Apparent weight of a body inside a liquid
If a body is completely immersed in a liquid, its effective weight gets decreased. The decrease in its weight is equal to the upthrust on the body.
Hence, apparent weight of body \(=w_{\text {app }}=w_{\text {actual }}-\) upthrust or
\(
w_{\mathrm{app}}=V \rho_s g-V \rho_l g
\)
Here, \(\quad V=\) total volume of the body,
\(\rho_s=\) density of body and \(\rho_l=\) density of liquid.
Thus, \(w_{\text {app }}=V g\left(\rho_s-\rho_l\right)\)
\(
\begin{array}{ll}
\text { or } & w_{\text {app }}=V \rho_s g\left(1-\frac{\rho_l}{\rho_s}\right) \\
\therefore & w_{\text {app }}=w_{\text {actual }}\left(1-\frac{\rho_l}{\rho_s}\right)
\end{array}
\)
Special cases Three possibilities may now arise
Case-I: If \(\rho_s<\rho_l\), then in this condition, the upthrust applied by the liquid will be greater than the weight of the body. That means, if the body is completely immersed in liquid, it will experience a net upward force. When released, the body comes up to the fluid surface till the upthrust becomes equal to the weight of the body, at this point, the body floats partially immersed in the fluid.

Case-II: If \(\rho_s=\rho_l\), then \(w_{\mathrm{app}}=0\) in this condition, upthrust of the liquid balances the weight of the body. The body floats completely submerged just below the surface of the fluid.

Case-III: If \(\rho_s>\rho_l\), then in this condition, the upthrust of the liquid is less than the weight of the liquid, i.e. it is not sufficient to balance the weight of the body, so the body sinks.

Example 19: (Density from Apparenet Weight of Fully Submerged Body) We tie a block of a material of density higher than the density of water to a thin thread and measure the tension in the thread as the block is lowered in water. When the block is in air, the tension is 50 N and when fully submerged, the tension is 40 N. What is the density of the material if the density of water is \(1000 \mathrm{~kg} / \mathrm{m}^3\).
Solution: Let \(W\) be the tension in air, and \(W^{\prime}\) the tension when fully submerged in water. Let \(V\) be the volume of the block, \(\rho_0\) the density of water, and \(\rho\) the density of the block.
From the two experiments we can write the following relations.
\(
\begin{aligned}
& W=V \rho g \\
& W^{\prime}=W-F_b \\
& F_b=V \rho_0 g
\end{aligned}
\)
The volume of the block can be written as
\(
V=\frac{W}{\rho g}
\)
Therefore,
\(
W^{\prime}=\frac{W}{\rho}\left(\rho-\rho_0\right) .
\)
Therefore,
\(
1-\frac{\rho_0}{\rho}=\frac{W^{\prime}}{W},
\)
which gives
\(
\rho=\frac{W}{W-W^{\prime}} \rho_0 .
\)
Now, putting in the numbers we get
\(
\rho=\frac{50}{10} \times 1000=5000 \mathrm{~kg} / \mathrm{m}^3 .
\)
Example 20: (Floating Steel Container in Water). A 1 -mm thick steel sheet is made into a cylindrical cup of outer radius 10 cm and height 30 cm with one end sealed and the other end open. It is then placed in water with the closed end first. How much of the cup stays above water? Data: density of water \(=1.0 \mathrm{~g} / \mathrm{cm}^3\), density of steel \(=8.0 \mathrm{~g} / \mathrm{cm}^3\).
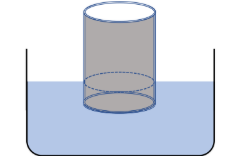
Solution: Let \(h\) be the height of the cylinder above water and \(H\) its total height. Let \(\boldsymbol{r}_{\text {out }}\) be the outer radius, \(\boldsymbol{r}_{\text {in }}\) the inner radius, and \(t\) the thickness. Let us denote the density of steel by \(\rho\) ad the density of water by \(\rho_0\).
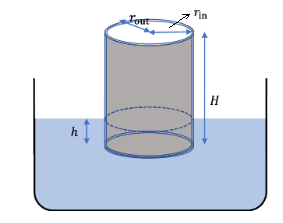
The force of buoyancy on the container will balance the weight of the container since the acceleration of the container is zero. To find an expression for the weight of the container, we figure out the volume of the material used to make the container by adding the volume of the side and the bottom.
\(
V=\left(\pi r_{\text {out }}^2-\pi r_{\text {in }}^2\right) H+\pi r_{\text {in }}^2 t .
\)
Therefore, weight of the container is
\(
W=\rho V g \dots(i)
\)
Recall the magnitude of the buoyancy force is equal to the weight of the displaced fluid. Since \(H-h\) of the height is under water, this gives the following for the magnitude of the buoyancy force.
\(
F_b=\rho_0\left[\pi r_{\text {out }}^2(H-h)\right] g \dots(ii)
\)
Equating \(W\) with \(F_b\) gives the equation,
\(
\rho_0\left[\pi r_{\text {out }}^2(H-h)\right] g=W,
\)
which can be solved for \(h\)
\(
h=\frac{W}{\rho_0 \pi r_{\text {out }}^2 g} .
\)
Putting in the full expression for \(W\) from Eq. (i), and setting \(r_{\text {out }}=R=r_{\text {in }}+t\)
\(
H-h=\frac{\rho}{\rho_0}[(1-x) H+x t],
\)
where
\(
x=\left(1-\frac{t}{R}\right)^2 .
\)
Now, we use the numerical values in cm units for length, we get
\(
x=\left(1-\frac{0.1}{10}\right)^2=0.99^2=0.9801
\)
Therefore, we get
\(
\begin{aligned}
H-h & =\frac{8.0}{1.0}[(1-0.9801) \times 30+0.9801 \times 0.1] \\
& =5.6 \mathrm{~cm}
\end{aligned}
\)
Hence,
\(
h=H-5.6 \mathrm{~cm}=24.4 \mathrm{~cm} .
\)
Example 21: Pushing Down a Ball Filled with Air into Water. A plastic spherical ball of negligible mass and filled with air has a radius of 10 cm . A boy pushes the ball in water till quarter of its volume is submerged. How much force the boy must apply to keep the ball so submerged? Assume negligible change in the volume of the ball.
Solution: Let \(\boldsymbol{F}\) be the force by the boy. This force on the ball balances the buoyancy force on the ball. We ignore the weight of the ball.
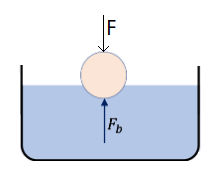
\(
F=F_b,
\)
with
\(
F_b=\frac{V}{4} \rho_w g .
\)
Therefore,we have
\(
\begin{aligned}
F & =\frac{1}{4} \frac{4}{3} \pi R^3 \rho_w g \\
& =\frac{1}{3} \pi \times 0.1^3 \times 10^3 \times 9.81=10.3 \mathrm{~N}
\end{aligned}
\)
The direction of this force is down.
Example 22: Apparent Weight of a Brass Block Half Submerged in Water:
A block of brass of mass 800 grams is attached to a string and hung from a support. The block is then placed so that half of the block is submerged in the water. What is the tension in the string?
Data: density of brass \(=8.6 \mathrm{~g} / \mathrm{cc}\), density of water \(=1.0 \mathrm{~g} / \mathrm{cc}\).
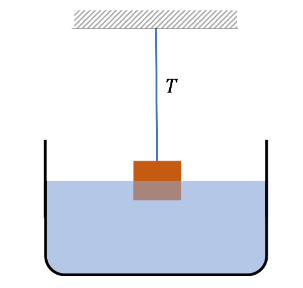
Solution: Let \(\rho\) denote the density of brass and \(\rho_0\) the density of water. There are three forces on the block: the weight \(M g\) of the block pointed down, the tension \(T\) in the string pointed up, and the buoyancy \(F_B\) pointed up. The forces are in balance.
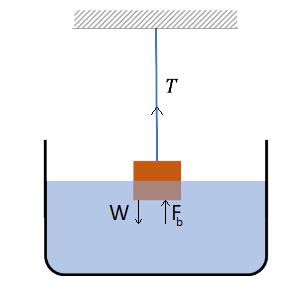
\(
F_b+T=M g .
\)
Half of the volume \(V\) of the block is in water, which we will write as \(V=M / \rho\). Therefore, the buoyancy is
\(
F_b=\frac{1}{2} V \rho_0 g=\frac{1}{2} \frac{M}{\rho} \rho_0 g
\)
The two equations give the magnitude of the tension in the string to be
\(
T=\left[1-\frac{1}{2} \frac{\rho_0}{\rho}\right] M g
\)
Now, putting in the numbers we get
\(
\begin{aligned}
T & =\left[1-\frac{1}{2} \times \frac{1.0 \mathrm{~g} / \mathrm{cc}}{8.6 \mathrm{~g} / \mathrm{cc}}\right] 0.8 \mathrm{~kg} \times 9.81 \mathrm{~m} / \mathrm{s}^2 \\
& =7.38 \mathrm{~N}
\end{aligned}
\)
Example 23: An ornament weighing 50 g in air weights only 46 g in water. Assuming that some copper is mixed with gold to prepare the ornament. Find the amount of copper in it. Specific gravity of gold is 20 and that of copper is 10 .
Solution: Let \(m\) be the mass of the copper in ornament.
Then, mass of gold in it is \((50-m)\).
Volume of copper, \(V_1=\frac{m}{10} \quad\left(\because \text { Volume }=\frac{\text { Mass }}{\text { Density }}\right)\)
and \(\quad\) volume of gold, \(V_2=\frac{50-m}{20}\)
When immersed in water ( \(\rho_w=1 \mathrm{gcm}^{-3}\) ),
decrease in weight \(=\) upthrust
\(
\therefore \quad(50-46) g=\left(V_1+V_2\right) \rho_w g
\)
\(
4=\frac{m}{10}+\frac{50-m}{20} \text { or } 80=2 m+50-m
\)
\(\therefore \quad m=30 \mathrm{~g}\)
Example 24: Density of ice is \(900 \mathrm{~kg} \mathrm{~m}^{-3}\). A piece of ice is floating in water (of density \(1000 \mathrm{~kg} \mathrm{~m}^{-3}\) ). Find the fraction of volume of the piece of ice outside the water.
Solution: Let \(V\) be the total volume and \(V_i\) the volume of ice piece immersed in water. For equilibrium of ice piece,
weight \(=\) upthrust
\(
\therefore \quad V \rho_i g=V_i \rho_w g \dots(i)
\)
Here,\(\quad \rho_i=\) density of ice \(=900 \mathrm{kgm}^{-3}\)
and \(\quad \rho_w=\) density of water \(=1000 \mathrm{kgm}^{-3}\)
Substituting in Eq. (i), we get
\(
\frac{V_i}{V}=\frac{\rho_i}{\rho_w}=\frac{900}{1000}=0.9
\)
i.e. The fraction of volume outside the water, \(f=1-0.9=0.1\)
Example 25: A piece of ice is floating in a glass vessel filled with water. How will the level of water in the vessel changes when the ice melts?
Solution: Let \(m\) be the mass of ice piece floating in water.
In equilibrium, weight of ice piece \(=\) upthrust
\(
m g=V_i \rho_w g \text { or } V_i=\frac{m}{\rho_w} \dots(i)
\)
Here, \(V_i\) is the volume of ice piece immersed in water.
When the ice melts, let \(V\) be the volume of water formed by \(m\) mass of ice.
Then,
\(
V=\frac{m}{\rho_w} \dots(ii)
\)
From Eqs. (i) and (ii), we see that, \(V_i=V\)
Hence, the level will not change.
Example 26: A piece of ice having a stone frozen in it, floats in a glass vessel filled with water. How will the level of water in the vessel changes when the ice melts?
Solution: Let \(m_1=\) mass of ice, \(m_2=\) mass of stone,
\(\rho_S=\) density of stone and \(\rho_w=\) density of water.
In equilibrium, when the piece of ice floats in water,
\(
\begin{array}{rlrl}
& \text { weight of (ice }+ \text { stone }) & =\text { upthrust } \\
\left(m_1+m_2\right) g & =V_i \rho_w g \dots(i) \\
\therefore \quad V_i & =\frac{m_1}{\rho_w}+\frac{m_2}{\rho_w}
\end{array}
\)
Here, \(V_i=\) volume of ice immersed.
When the ice melts, \(m_1\) mass of ice converts into water and stone of mass \(m_2\) is completely submerged.
Volume of water formed by \(m_1\) mass of ice, \(V_1=\frac{m_1}{\rho_w}\)
Volume of stone (which is also equal to the volume of water displaced),
\(
V_2=\frac{m_2}{\rho_S}
\)
Since, \(\rho_S>\rho_w\), so \(V_1+V_2<V_i\) or the level of water will decrease.
Buoyant force in accelerating fluids
If a body is dipped inside a liquid of density \(\rho_L\) placed in an elevator moving upward with an acceleration \(a\) as shown in figure.

Now, consider the force acting on the liquid replaced by the body. For upward motion of the replaced liquid, we can write,
\(
F-w=m a
\)

where, \(w=\) weight of the displaced liquid \(F=\) buoyant force acting on the bodly
\(\therefore \quad F=w+m a=m(g+a)=\rho_L V(g+a)\)
\(
F=V \rho_L g_{\mathrm{eff}}
\)
Here, \(g_{\mathrm{eff}}=|g+a|\)
e.g. If the lift is moving upwards with an acceleration \(a\), the value of \(g_{\text {eff }}\) is \(g+a\) and if it is moving downwards with acceleration \(a\), the \(g_{\text {eff }}\) is \(g-a\). In a freely falling lift, \(\boldsymbol{g}_{\text {eff }}\) is zero (as \(a=g\) ) and hence, net buoyant force is zero. This is why, in a freely falling vessel filled with some liquid, the air bubbles do not rise up (which otherwise move up due to buoyant force).
Example 27: The tension in a string holding a solid block below the surface of a liquid (of density greater than that of solid) as shown in figure is \(T_0\) when the system is at rest. What will be the tension \(T\) in the string, if the system has an upward acceleration \(a\)?

Solution: Let \(m\) be the mass of block.
Initially for the equilibrium of block, \(F=T_0+m g \dots(i)\)

Here, \(F\) is the upthrust on the block.
When the lift is accelerated upwards, \(g_{\text {eff }}\) becomes \(g+a\) instead of \(g\).
Hence, \(\frac{F^{\prime}}{F}=\frac{m(g+a)}{mg}\)
\(
F^{\prime}=F\left(\frac{g+a}{g}\right) \dots(ii)
\)
From Newton’s second law,
\(
F^{\prime}-T-m g=m a \dots(iii)
\)
Solving Eqs. (i), (ii) and (iii), we get
\(
T=T_0\left(1+\frac{a}{g}\right)
\)
Example 28: A 700 g solid cube having an edge of length 10 cm floats in water. How much volume of the cube is outside the water? Density of water \(=1000 \mathrm{~kg} \mathrm{~m}^{-3}\).
Solution: The weight of the cube is balanced by the buoyant force. The buoyant force is equal to the weight of the water displaced. If a volume \(V\) of the cube is inside the water, the weight of the displaced water \(=V \rho g\), where \(\rho\) is the density of water. Thus,
\(
V \rho g=(0 \cdot 7 \mathrm{~kg}) g
\)
or, \(\quad V=\frac{0 \cdot 7 \mathrm{~kg}}{\rho}=\frac{0 \cdot 7 \mathrm{~kg}}{1000 \mathrm{~kg} \mathrm{~m}^{-3}}=7 \times 10^{-4} \mathrm{~m}^3=700 \mathrm{~cm}^3\).
The total volume of the cube \(=(10 \mathrm{~cm})^3=1000 \mathrm{~cm}^3\).
The volume outside the water is
\(
1000 \mathrm{~cm}^3-700 \mathrm{~cm}^3=300 \mathrm{~cm}^3 .
\)