1.1 Introduction
What is Physics?
Physics is the scientific study of matter, energy, and the fundamental forces that govern their interactions, from the smallest subatomic particles to the entire universe. It uses mathematical principles and experimentation to explain how things move, why they have certain properties, and the cause-and-effect relationships in the natural world. Everyday examples include using the force of gravity to predict how high a ball will travel when thrown and understanding how electricity from a battery powers a car engine.
Examples of physics in action:
- Mechanics: This branch studies motion and its causes. When you throw a ball, physics explains how gravity affects its arc and how you can calculate its trajectory using algebra.
- Electromagnetism: This deals with electricity, magnetism, and their relationship. A car battery stores electrical energy, which is then used to start the engine and power the lights and radio, illustrating the principles of electromagnetism.
- Energy: Physics explores how energy is transferred and transformed. The energy from a falling tree is converted into sound and kinetic energy as it hits the ground, and the energy from a battery is converted into mechanical energy to run a clock.
- Thermodynamics: This area of physics is concerned with heat and temperature. When you feel the warmth of a cup of coffee, you are experiencing the transfer of thermal energy from the coffee to your hand.
- Quantum Mechanics: This field studies the behavior of matter and energy at the atomic and subatomic levels. It is the underlying principle for modern technologies like transistors and lasers, which are used in everything from smartphones to medical equipment.
What is Matter in Physics?
In physics, matter is any substance that has mass and occupies space, meaning it has volume. It is the “stuff” that makes up the universe and can be found in various states, such as solid, liquid, and gas. Matter is ultimately composed of atoms, which are made of subatomic particles like protons, neutrons, and electrons.
Key characteristics and composition
- Mass and volume: The defining properties of matter are that it has mass and takes up space (has volume).
- Atomic structure: All matter is made of atoms, which are in turn composed of smaller particles.
- Subatomic particles: Atoms consist of protons, neutrons, and electrons. At an even more fundamental level, quarks and leptons are the elementary particles that make up protons and neutrons.
States of matter
- Solid: Maintains a definite shape and volume.
- Liquid: Takes the shape of its container but has a definite volume.
- Gas: Lacks both a definite shape and volume, expanding to fill its container.
- Plasma: A fourth state of matter, often referred to as ionized gas.
What is not matter
- Massless particles, such as photons, are not considered matter.
- Energy, like heat and light, is distinct from matter.
Units
A “unit” is a standard for measuring a quantity, such as length, mass, or time. The result of a measurement of a physical quantity is expressed by a number (or numerical measure) accompanied by a unit. In measurement a standard amount of a physical quantity used for comparison.
Examples include:
- Length: meters, feet, inches
- Mass: kilograms, grams
- Time: seconds, hours
Physical Quantities
“Anything which we can measure is called physical quantity.”
For example,
\begin{array}{cl}
\text { Temperature }=40^{\circ} \mathrm{C} & \longrightarrow \text { Physical quantity. } \\
\text { Length }=30 \mathrm{~m} & \longrightarrow \text { Physical quantity. } \\
\text { Time }=5 \mathrm{~min} & \longrightarrow \text { Physical quantity. } \\
\text { Mass }=2 \mathrm{~kg} & \longrightarrow \text { Physical quantity. } \\
\text { Happiness }=? & \longrightarrow \text { Not a physical quantity. } \\
\text { Sadness }=? & \longrightarrow \text { Not a physical quantity. }
\end{array}
\)

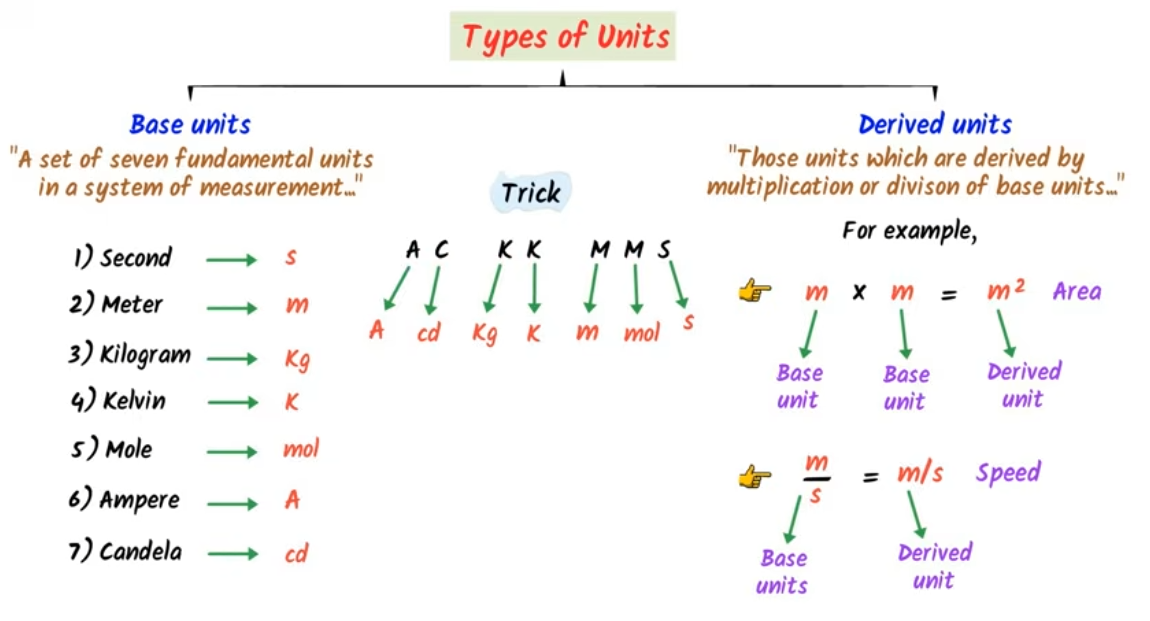
Fundamental (base) quantities and Fundamental (base) Units
Physical quantities which do not depend upon other quantities for their complete definition are known as fundamental or base quantities. The units of these quantities are called fundamental or base units. e.g. length, mass, time, temperature, electric current, amount of substance, and luminous intensity, etc. a unit speed. Table below shows Fundamental or Base Quantities:
Fundamental or Base Quantities:
\(\begin{array}{lccc}
\text { Quantity } & \text { Name of the Unit } & & \text { Symbol } \\
\text { Length } & \text { metre } & & \mathrm{m} \\
\text { Mass } & \text { kilogram } & & \mathrm{kg} \\
\text { Time } & \text { second } & & \mathrm{s} \\
\text { Electric Current } & \text { ampere } & & \mathrm{A} \\
\text { Thermodynamic Temperature } & \text { kelvin } & & \mathrm{K} \\
\text { Amount of Substance } & \text { mole } & & \mathrm{mol} \\
\text { Luminous Intensity } & \text { candela } & & \mathrm{cd}
\end{array}
\)
Derived quantities and derived units
The quantities which can be expressed in terms of the fundamental quantities are called derived quantities. The units of these quantities are called derived units.
e.g. Unit of speed \(=\mathrm{ms}^{-1}\) can be derived from fundamental units, i.e. unit of length and time as
\(
\text { Speed }=\frac{\text { Distance }}{\text { Time }}=\frac{\mathrm{m}}{\mathrm{~s}}=\mathrm{ms}^{-1}
\)
Other examples of derived quantities are: volume, velocity, acceleration, force, pressure, momentum, energy, etc.