1.3 Significant figures
Order of magnitude
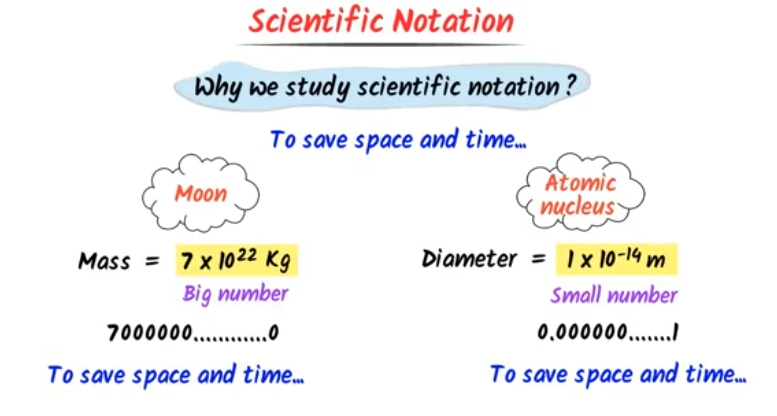
Any physical quantity can be expressed in the form of \(a \times 10^b\) (in terms of magnitude), where \(a\) is a number lying between 1 and 10 ; and \(b\) is any negative or positive exponent of 10, then the exponent \(b\) is called the order of magnitude of the physical quantity. And the expression of a quantity as \(a \times 10^b\) is called scientific notation.
e.g. The speed of light is given as \(3.00 \times 10^8 \mathrm{~m} / \mathrm{s}\). So, the order of magnitude of the speed of light is 8.
The order of magnitude gives an estimate of the magnitude of the quantity. The charge on an electron is \(16 \times 10^{-19} \mathrm{C}\). Therefore, we can say that the charge possessed by an electron is of the order \(10^{-19}\) or its order of magnitude is -19.
Significant Figures
The significant figures are normally those digits in a measured quantity which are known reliable or about which we have confidence in our measurement plus one additional digit that is uncertain. e.g. If length of some object is 185.2 cm , then it has four significant figures. The digits 1,8 and 5 are reliable and digit 2 is uncertain.
Note: Significant figures indicate the precision of the measurement which depends on the least count of the measuring instrument.


Rules for rounding off digits:
- If the digit to the right of the last reported digit is less than 5 round it and all digits to its right off.
- If the digit to the right of the last reported digit is greater than 5 round it and all digits to its right off and increased the last reported digit by one.
- If the digit to the right of the last reported digit is a 5 followed by either no other digits or all zeros, round it and all digits to its right off, and if the last reported digit is odd round up to the next even digit. If the last reported digit is even then leave it as is.
For example, if we wish to round off the following number to 3 significant digits : \(18.3682\), the answer is 18.4. Another example: Round off \(4.565\) to three significant digits, the answer would be \(4.56\).
Rounding off
\(
\begin{array}{ccc}
6.87 \rightarrow 6.9, & 6.84 \rightarrow 6.8, & 6.85 \rightarrow 6.8, \\
6.75 \rightarrow 6.8, & 6.65 \rightarrow 6.6, & 6.95 \rightarrow 7.0
\end{array}
\)
Example 1: How many significant figures are there in the following measured values?
(i) 227.2 g
(ii) 3600 g
(iii) 0.00602 g
(iv) \(2.50 \times 10^{10} g\)
Solution: (i) 227.2 g has all the non-zero digits. Hence, it has four significant figures.
(ii) According to rule number 4, trailing zeros are not significant. Hence, 3600 g has 2 significant figures.
(iii) According to the rule number 3, the zeros on the right of decimal point but to the left of first non-zero digit are not significant. Hence, 0.00602 g has 3 significant figures.
(iv) According to the rule number 6, it has 3 significant figures.
Mathematical operations of significant figures
The result of a mathematical operation involving measured values of quantities cannot be more accurate than the measured value themselves. So, certain rules have to be followed while doing mathematical operations with significant figures, so that precision in final result is consistent with the precision of the original measured values.
Addition or subtraction
Suppose in the measured values to be added or subtracted, the least number of significant digits after the decimal is \(n\). Then, in the sum or difference also, the number of significant digits after the decimal should be \(n\).
\(
\text { e.g. } 1.2+3.45+6.789=11.439 \approx 11.4
\)
Here, the least number of significant digits after the decimal is one. Hence, the result will be 11.4 (when rounded off to smallest number of decimal places).
Similarly, e.g. \(12.63-10.2=2.43 \approx 2.4\)
Example2: Add \(6.75 \times 10^3 \mathrm{~cm}\) to \(4.52 \times 10^2 \mathrm{~cm}\) with regard to significant figures.
Solution: Let \(a=6.75 \times 10^3 \mathrm{~cm}, b=4.52 \times 10^2 \mathrm{~cm}\)
\(
=0.452 \times 10^3 \mathrm{~cm}=0.45 \times 10^3 \mathrm{~cm} \text { (upto 2 places of decimal) }
\)
∴ Addition of significant figures
\(
a+b=\left(6.75 \times 10^3+0.45 \times 10^3\right) \mathrm{cm}=7.20 \times 10^3 \mathrm{~cm}
\)
Example 3: Two sticks of lengths 12.132 cm and 10.2 cm are placed end to end. Find their total length with due regard to significant figures.
Solution: Length of first stick \(=12.132 \mathrm{~cm}\) (5 significant figures)
Length of second stick \(=10.2 \mathrm{~cm} \quad\) (3 significant figures)
∴ Total length of two sticks \(=12.132+10.2=22.332\)
The answer should be rounded off with least number of significant digits after the decimal.
∴ Total length of two sticks will be 22.3 cm.
Multiplication or division
Suppose in the measured values to be multiplied or divided, the least number of significant digits be \(n\), then in the product or quotient, the number of significant digits should also be \(n\). e.g. \(1.2 \times 36.72=44.064 \approx 44\)
The least number of significant digits in the measured values are two. Hence, the result when rounded off to two significant digits become 44 . Therefore, the answer is 44.
Similarly, e.g. \(\frac{1100}{10.2}=107.8431373 \approx 110\)
As 1100 has minimum number of significant figures (i.e. 2), therefore the result should also contain only two significant digits. Hence, the result when rounded off to two significant digits becomes 110.
Example 4: The voltage across a lamp is 6.32 V when the current passing through it is 3.4 A. Find the power consumed upto appropriate significant figures.
Solution: Voltage across a lamp, \(V=6.32 \mathrm{~V}\) (3 significant figures) Current flowing through lamp, \(I=3.4 \mathrm{~A}\) (2 significant figures)
∴ Power consumed, \(P=V I=(6.32)(3.4)=21.488 \mathrm{~W}\)
Answer should have minimum number of significant figures. Here, the minimum number of significant figures is 2.
∴ Power consumed \(=21 \mathrm{~W}\)
Example 5: A thin wire has a length of 21.7 cm and radius 0.46 mm . Calculate the volume of the wire upto correct significant figures.
Solution: Given, \(l=21.7 \mathrm{~cm}, r=0.46 \mathrm{~mm}=0.046 \mathrm{~cm}\)
Volume of wire, \(V=\pi r^2 l=\frac{22}{7}(0.046)^2(21.7)\)
\(
=0.1443 \mathrm{~cm}^3 \simeq 0.14 \mathrm{~cm}^3
\)
Example 6: The time taken by a pendulum to complete 25 vibrations is 88.0 s. Find the time period of the pendulum in seconds upto appropriate significant figures.
Solution:
\(
\begin{aligned}
\text { Time period of oscillation } & =\frac{\text { Total time taken }}{\text { Number of oscillations }} \\
& =\frac{88.0}{25} \mathrm{~s}=3.52 \mathrm{~s}
\end{aligned}
\)
Out of the two quantities given in the data, 25 is exact, hence has infinite significant figures. Therefore, the answer should be reported to three significant figures, i.e. 3.52 s.
Example 7: 5.74 g of substance occupies \(1.2 \mathrm{~cm}^3\). Express its density by keeping the significant figures in view.
Solution: Here, mass, \(m=5.74 \mathrm{~g}\), volume, \(V=1.2 \mathrm{~cm}^3\) As density, \(\quad \rho=\frac{\text { mass }}{\text { volume }}=\frac{5.74 \mathrm{~g}}{1.2 \mathrm{~cm}^3}=4.783 \mathrm{~g} \mathrm{~cm}^{-3}\)
As mass has 3 significant digits and volume has 2 significant digits, therefore as per rule, density will have only two significant digits, rounding off, we get \(\rho=4.8 \mathrm{gcm}^{-3}\).